Product Images Xopenex
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 20 images provide visual information about the product associated with Xopenex NDC 17478-174 by Akorn, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
xop07 0005 06
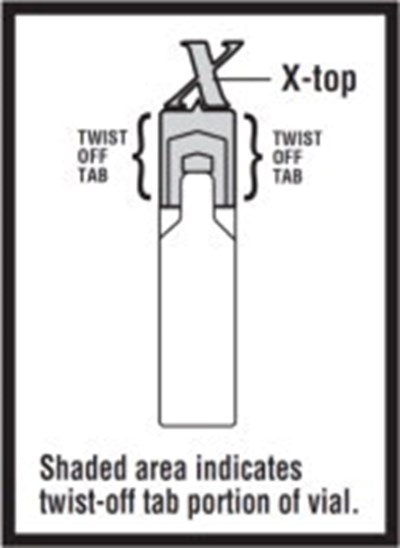
The text states "Shaded area indicates twist-off tab portion of vial." It may be a label or instruction on a vial indicating where to twist to open it.*
xop07 0005 15

This is a pharmaceutical product containing levalbuterol hydrochloride used for inhalation. The medication is provided in unit-dose vials, and each vial contains 0.31 mg of the medication. This inhalation solution should be used as directed by a physician and should not be used beyond the recommended dosage. The medication must be protected from light and stored at a controlled room temperature of 20 to 25°C (68° to 77°F). The individual vials should be used within one week of being removed from the protective foil pouch. Once the foil pouch is opened, the unit-dose vials should be used within two weeks.*
xop07 0005 17

This is a medical product information leaflet for AKN1024-801R00 containing the NDC code, description of potency, storage instructions, and directions for use. It is a unit-dose vial used to treat bronchospasm and is to be stored in a foil pouch at a controlled room temperature between 20° to 25°C. The pharmacist must detach the patient instructions for use before dispensing, and it is not recommended to exceed the recommended dosage. The solution in the vials should be colourless, and the vials should be discarded if not used within two weeks after opening the foil pouch.*
xop07 0005 19

This is a product description for a medication called Xopenex (levalbuterol HCl) Inhalation Solution. Each unit dose vial contains 1.25 mg of levalbuterol in an aqueous solution. The solution is free of preservatives and should be used as directed by a physician. The vials should be stored in the protective foil pouch and used within two weeks of opening. The medication should be discarded if the solution is not colorless. The product is distributed by Akon, Inc. for Oak Pharmaceuticals Inc.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.















