Product Images Cromolyn Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 8 images provide visual information about the product associated with Cromolyn Sodium NDC 17478-291 by Akorn, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
cro03 0004 07

This is a 10mL sterile ophthalmic solution containing Cromolyn Sodium 40mg/mL (4%) as the active ingredient, with Edetate Disodium (0.1%) and Water for Injection as the inactive ingredients. The solution should be stored between 20° to 25°C (68° to 77°F), and must be protected from light. The recommended dosage for ophthalmic use is 1-2 drops in each eye, 4 to 6 times a day at regular intervals, as indicated in the package insert. The solution is preserved with Benzalkonium Chloride (0.01%). This solution is for ophthalmic use only.*
cro03 0004 08
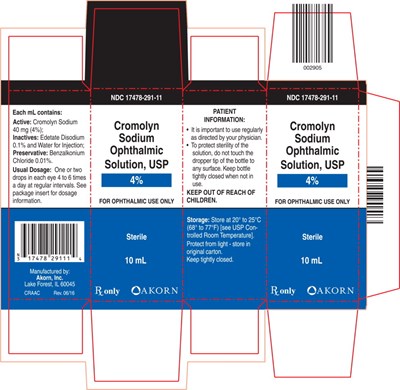
Cromolyn Sodium Ophthalmic Solution, USP is a prescription medicine used for ophthalmic purposes only. It is manufactured by Akorn, Inc. and contains 40mg (4%) Cromolyn Sodium, 0.1% Edetate Disodium Sodium and water for injection. Additionally, it contains 0.01% Benzalkonium Chloride as a preservative. The recommended dosage is one or two drops applied in each eye 4 to 6 times per day, as prescribed by a doctor. Patients must not touch the dropper tip to any surface and store the bottle in its original carton at tightly closed conditions, away from light and children. The package contains 10mL Cromolyn Sodium Ophthalmic Solution, USP. Detailed dosage information is available in the package insert.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





