Product Images Namenda
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Namenda NDC 21695-169 by Rebel Distributors Corp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image of Figure 2: Cumalative percentage graph - 74ce605d b493 4b95 9d59 9253f81f4e20 03

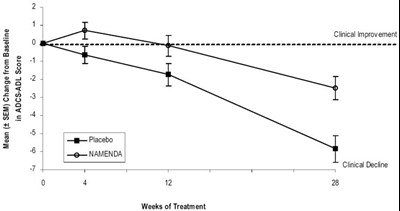
The text is a graph displaying the cumulative percentage of patients on Placebo and NAMENDA, with respect to their ADCS-ADL change from baseline. The x-axis displays the change from the baseline (from -5 to 25), while the y-axis displays the cumulative percentage of patients.*
image of Figure 3: Time course of the change graph - 74ce605d b493 4b95 9d59 9253f81f4e20 04
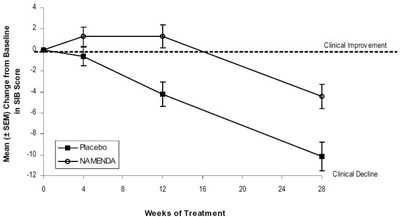
The text refers to a graph or table representing the SIB score, a measurement of cognitive function, and the mean change from baseline after treatment with either placebo or Namenda over a period of 16 weeks. There is a gap in the information provided for the 6 weeks between the 12 and 16 marks. The graph or table also includes information on clinical decline.*
image of Figure 4: Cumulative percentage graph - 74ce605d b493 4b95 9d59 9253f81f4e20 05

The text appears to be a graph or chart displaying the cumulative percentage of patients for two different treatments, Placebo and NAMENDA. The graph also includes a line graph representing the change from baseline in SIB (Severe Impairment Battery) scores. The x-axis shows the percentage, while the y-axis shows the change from baseline. There is not enough information to provide further details.*
image of Figure 5: Time course of the change graph - 74ce605d b493 4b95 9d59 9253f81f4e20 06

This is a graph showing the mean change from baseline in ADCS-ADL score in patients receiving either placebo/donepezil or Namenda/donepezil. The graph shows that patients who received Namenda/donepezil had less clinical decline compared to those who received placebo/donepezil. The graph also shows the weeks of treatment (0-18) and the mean score values.*
image of Figure 6: Cumulative percentage graph - 74ce605d b493 4b95 9d59 9253f81f4e20 07

This text represents a graph showing the cumulative percentage of patients with Alzheimer's disease and the level of improvement on an activity of daily living scale (ADCS-ADL) after treatment with either placebo/donepezil or NAVENDA/donepezil. The graph indicates that a higher percentage of patients experience a positive change in their ADCS-ADL score after treatment with NAVENDA/donepezil compared to placebo/donepezil.*
image of Figure 7: Time course of the change graph - 74ce605d b493 4b95 9d59 9253f81f4e20 08

The text describes a clinical improvement in the SIB score, with a graph displaying the mean change from the baseline for a placebo and a medication called NAMENDA/Donep. The x-axis shows the weeks of treatment, and the y-axis displays the SIB score. There is also a line indicating clinical decline.*
image of Figure 8: Cumulative percentage graph - 74ce605d b493 4b95 9d59 9253f81f4e20 09

This is a graph that compares the cumulative percentage of patients and their SIB (Severe Impairment Battery) change from baseline when taking either a placebo and Donepezil combination or Namenda and Donepezil combination. The graph shows that the Namenda and Donepezil combination group has a better SIB change from baseline than the placebo and Donepezil group.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.