Product Images Lyrica
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 19 images provide visual information about the product associated with Lyrica NDC 21695-663 by Rebel Distributors Corp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 01
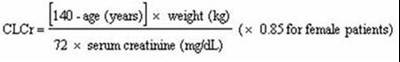
This text appears to be a medical formula for estimating the glomerular filtration rate (GFR) in adults using the serum creatinine level, age, weight, and gender. However, because some characters are not readable, it is difficult to provide an accurate description.*
Figure - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 04

This is a table that shows the percentage of patients improved (labeled as "Percent Improvement") from a baseline. However, it is not clear what the letters and numbers in the middle of the table represent.*
Figure - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 07
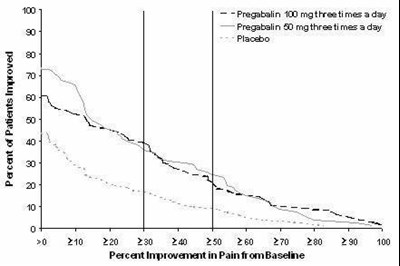
This appears to be a chart showing the percentage improvement in pain from baseline for patients receiving Pregatalin 100mg three times per day, Pregabalin 0mg three times per day, and placebo. However, the chart is not entirely readable due to character recognition errors, so some details may be missing or inaccurate.*
Logo - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 12

This is a description of a product or medication that is distributed by Parke-Davis, a division of Pfizer Inc located in NY, NY (zip code 10017).*
Logo - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 13

This is a text that indicates the distribution company of a product. The company is named Parke-Davis and it is a division of Pfizer Inc located in NY, NY 10017.*
Lyrica 25mg - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 14

Lyrica is a medication available in the form of capsules containing 26mg size. Each capsule contains 25mg of Pregabalin and this medication is identified with the product ID PLO27615.*
Lyrica 75mg - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 15

This is a description of a medication called Lyrica manufactured by Pfizer Pharmaceuticals LLC. It comes in hard gelatin capsules printed with "Pfizer" on the cap and "PGN 75" on the body. Each capsule contains 75mg of Pregabalin, and the product ID is PL027872. The medication is stored at controlled room temperature (15°C-30°C), and it is important to keep it out of the reach of children. The text also indicates that Physician Partner in Thousand Oaks, CA packaged the medication.*
Lyrica 100mg - 8fb45a78 b2d8 45dd b844 0de21d1fdde9 17
This is a medication description. The name of the medication is not available as the has scanned it incorrectly. The medication comes in the form of capsules, each containing 150mg. The producer of the medication is Pfizer Pharmaceuticals LLC and the location mentioned is Vega Baja, Puerto Rico with the product ID PL0O86230.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.










