Product Images Losortan Potassium
View Photos of Packaging, Labels & Appearance
- Structured formula for losartan - 41a61cf1 30a3 472b a592 b17a610daaba 01
- Structured formula for losartan - 41a61cf1 30a3 472b a592 b17a610daaba 02
- Structured formula for losartan - 41a61cf1 30a3 472b a592 b17a610daaba 03
- Structured formula for losartan - 41a61cf1 30a3 472b a592 b17a610daaba 04
- Structure product formula for Losartan - 41a61cf1 30a3 472b a592 b17a610daaba 05
- Losartan Potassium 25mg - 41a61cf1 30a3 472b a592 b17a610daaba 06
Product Label Images
The following 6 images provide visual information about the product associated with Losortan Potassium NDC 21695-989 by Rebel Distributors Corp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Structured formula for losartan - 41a61cf1 30a3 472b a592 b17a610daaba 02
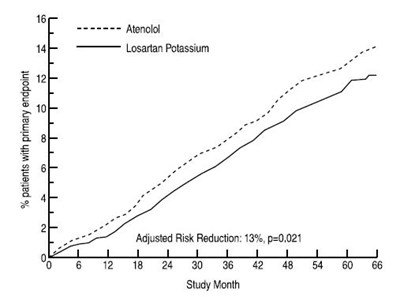
This appears to be a portion of a clinical study report comparing the efficacy of Atenolol and Losartan Potassium in reducing risk in patients with a primary endpoint. The adjusted risk reduction was found to be 13% with a p-value of 0.021. The table underneath shows the study month with corresponding numbers that are not clearly explained.*
Structure product formula for Losartan - 41a61cf1 30a3 472b a592 b17a610daaba 05
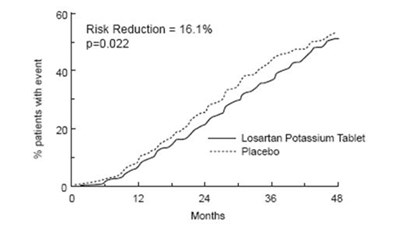
This is a clinical trial result suggesting that only 9% of patients experienced a ventricular tachycardia (vt) event while taking Losartan Potassium Tablet compared to those taking a placebo. Risk reduction was found to be 16.1% with a statistical significance of p=0.022. However, the rest of the text is not decipherable.*
Losartan Potassium 25mg - 41a61cf1 30a3 472b a592 b17a610daaba 06
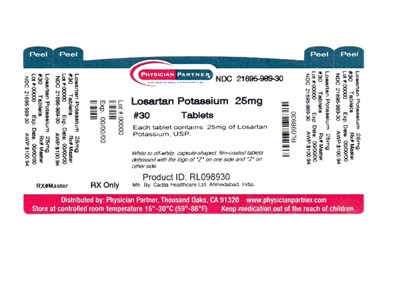
This is the description of a medication called Losartan Potassium 25mg. It comes in a bottle of 30 tablets that are white, capsule-shaped, and film-coated. The tablets have the number "2" on one side and "6" on the other. The medication is produced by Cadia Healthcare Ltd. in Ahmedabad, India, and the product ID is RL098930. The storage temperature for the medication is between 59°F and 86°F. A caution to keep the medication out of reach of children is also mentioned.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


