FDA Label for Purell Waterless Surgical Scrub
View Indications, Usage & Precautions
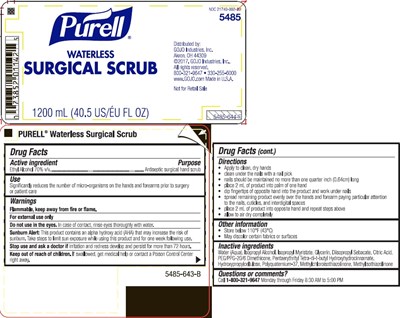
Purell Waterless Surgical Scrub Product Label
The following document was submitted to the FDA by the labeler of this product Gojo Industries, Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
Ethyl alcohol 70% v/v
Purpose
Antiseptic surgical hand scrub
Uses
Significantly reduces the number of micro-organisms on the hands and forearms prior to surgery or patient care
Warnings
Flammable. Keep away from fire or flame.
For external use only
Sunburn Alert: This product contains an alpha hydroxy acid (AHA) that may increase the risk of sunburn. Take steps to limit sun exposure while using this product and for one week following use.
Otc - When Using
Do not use in the eyes. In case of contact, rinse eyes thoroughly with water.
Otc - Stop Use
Stop use and ask a doctor if irritation and redness develop and persist for more than 72 hours.
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. If swallowed, get medical help or contact a Poison Control Center right away.
Directions
- clean under the nails with a nail pick
- mails should be maintained with a 1 millimeter free edge
- place 2 mL of product into palm of one hand
- dip fingers of opposite hand into the product and work under nails
- spread remaining product evenly over the hands and lower 2/3 of one forearm paying particular attention to the nails, cuticles, and interdigital spaces
- place 2 mL of product into opposite hand and repeat steps above
- allow to air dry completely
Inactive Ingredients
Water (Aqua), Isopropyl Myristate, Glycerin, Diisopropyl Sebacate, Citric Acid, PEG/PPG-20/6 Dimethicone, Tetradibutyl Pentaerythrityl Hydroxyhydrocinnamate, Hydroxypropylcellulose, Polyquaternium-37, Methylchloroisothiazolinone, Methylisothiazolinone
Package Label.Principal Display Panel
* Please review the disclaimer below.