Product Images Glipizide And Metformin Hcl
View Photos of Packaging, Labels & Appearance
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 01
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 02
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 03
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 04
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 05
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 06
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 07
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 08
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 09
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 10
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 11
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 12
- ee5fdf68 df2d 4965 8224 2c2df30f2f22 13
Product Label Images
The following 13 images provide visual information about the product associated with Glipizide And Metformin Hcl NDC 23155-117 by Heritage Pharmaceuticals Inc. D/b/a Avet Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
ee5fdf68 df2d 4965 8224 2c2df30f2f22 03
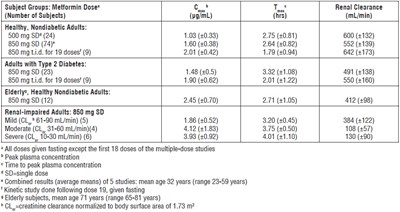
This is a table showing the Metformin Dose vs Renal Clearance, including the number of subjects, peak plasma concentration, time to peak plasma concentration, and creatinine clearance of healthy adults (both diabetic and non-diabetic), elderly and renal-impaired adults. The data suggests that the dose should be adjusted based on the patient's renal function.*
ee5fdf68 df2d 4965 8224 2c2df30f2f22 04
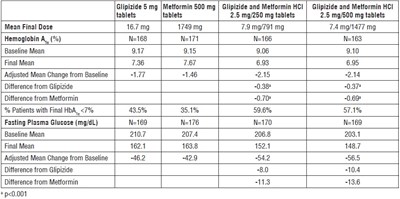
This is a table showing the dosage and efficacy of Glipizide and Metformin HCI tablets in treating patients with type 2 diabetes. The table includes information on mean final doses, Hemoglobin A1c levels, and fasting plasma glucose levels for various dosages of the medication. The table also includes differences in efficacy compared to Glipizide and Metformin alone, as well as the percentage of patients who achieved a final Hemoglobin A1c level below 7%.*
ee5fdf68 df2d 4965 8224 2c2df30f2f22 05

This is a comparison of Glipizide, Metformin, and Glipizide and Metformin HCI. The table shows mean final dose, Hemoglobin A and % of patients with HbA__<7%, Fasting Plasma Glucose, and their differences from each other. Adjusted Mean Change from Baseline is also shown. The results of the comparison are statistically significant with p<0.001.*
ee5fdf68 df2d 4965 8224 2c2df30f2f22 06

This is a table showing the number and percentage of patients with various adverse events related to Glipizide and Metformin HCI in different tablet strengths (2.5 mg/250 mg and 2.5 mg/500 mg) as well as Glipizide 5mg and Metformin 500mg tablets. Adverse events include upper respiratory infection, diarrhea, dizziness, hypertension, nausea, and vomiting. No additional context or information is available.*
ee5fdf68 df2d 4965 8224 2c2df30f2f22 07

This appears to be a table showing the incidence of various adverse events in patients taking different types of tablets - Glipizide 5 mg tablets, Metformin 500 mg tablets, and Glipizide and Metformin HCI tablets. The number of patients for each type of tablet is listed, followed by the percentage of those patients who experienced each adverse event. Some of the adverse events listed include diarrhea, headache, upper respiratory infection, musculoskeletal pain, and nausea/vomiting. It also notes that the dose of glipizide was fixed at 30 mg daily, while doses of metformin and Glipizide and Metformin HCI Tablets were titrated.*
ee5fdf68 df2d 4965 8224 2c2df30f2f22 08

Glipizide and Metformin HCI Tablets USP is a medication used to treat type 2 diabetes. It is available in different strengths and quantities in a bottle. The product is identified by the NDC code 23155-xxx-xx. The specific codes for different strengths and quantities are listed in the table.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.