Product Images Neostigmine Methylsulfate
View Photos of Packaging, Labels & Appearance
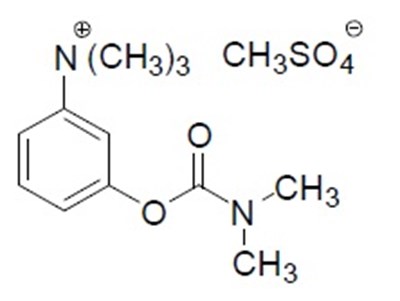
- structure - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 01
- logo - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 02
- label 10mg - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 03
- 10 mg monocarton - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 04
- 10 mg carton for 10 vial - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 05
- 5 mg label - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 06
- 5 mg carton - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 07
- 0.5 mg carton for 10 - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 08
Product Label Images
The following 8 images provide visual information about the product associated with Neostigmine Methylsulfate NDC 23155-517 by Heritage Pharmaceuticals Inc. D/b/a Avet Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
label 10mg - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 03

Neostigmine Methylsulfate Injection, USP is a prescription drug with NDC number 23155-518-31. The drug is available in 10 mg/10mL concentration for intravenous use and comes in a 10 mL multiple dose vial. It contains Neostigmine Methylsulfate USP 1mg, Phenol USP 4.5 mg, Sodium Acetate Trihydrate USP 0.2 mg, and Water for Injection USP. The pH is adjusted with Sodium Hydroxide NF/Acetic Acid USP. The drug should be stored in a carton and protected from light at 20° to 25°C (68° to 77°F) with excursions permitted between 15° to 30°C (59° to 86°F) as indicated by USP Controlled Room Temperature. The manufacturer of the drug is Emcure Pharmaceuticals Ltd., Sanand, Ahmedabad - 382110, India, and it is manufactured for Avet Pharmaceuticals Inc.*
10 mg monocarton - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 04
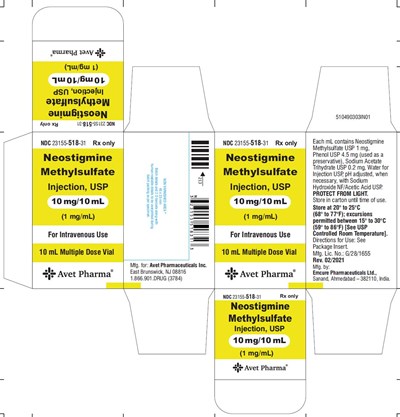
This is a label for a medication called Neostigmine Methylsulfate Injection, USP. It is a drug used intravenously and comes in a 10 mL multiple-dose vial. It contains Neostigmine Methylsulfate USP 1 mg/mL, Phenol USP 4.5 mg, Sodium Acetate Trihydrate USP 0.2 mg, and Water for Injection USP, preserved with Phenol. The instructions for use are on the package insert. The medication is produced by Avet Pharmaceuticals Inc. and manufactured by Emcure Pharmaceuticals Ltd. in India. The label also includes information about storage and handling of the medication.*
10 mg carton for 10 vial - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 05
Neostigmine Methylsulfate is a medicine with a concentration of 10mg/10mL, intended for injection. The information contains several errors and inconsistencies.*
5 mg label - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 06

Neostigmine Methylsulfate USP Injection, USP is a prescription drug contained in a vial with 5mg/10mL strength for intravenous use. The vial contains 10 mL of the solution that has Phenol USP 4.5 mg as a preservative, Sodium Acetate Trihydrate USP 0.2 mg, Water for Injection USP, pH adjusted, when necessary, with Sodium Hydroxide NF/Acetic Acid USP. The solution must be protected from light and stored in a carton. The storing temperature is between 20° to 25°C (68° to 77°F) with excursions permitted between 15° to 30°C (59° to 86°F). The manufacturer of the drug is Emcure Pharmaceuticals Ltd., Sanand, Ahmedabad - 382110, India, and Avet Pharmaceuticals Inc. manufactures it.*
5 mg carton - 719fb14c fc9a 46c9 a7e3 ca4c8a4a8525 07
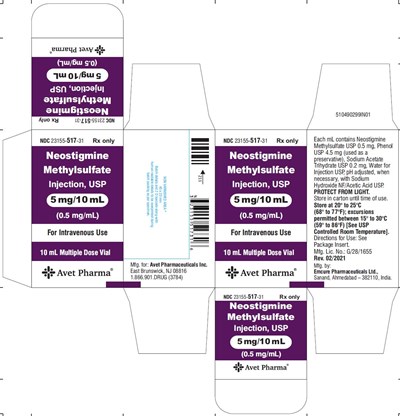
Neostigmine Methylsulfate Injection, USP is a medication that comes in a 10 mL multiple-dose vial, each containing 5mg of neostigmine methylsulfate USP in 10 mL of solution. This medication is intended for intravenous use and is manufactured by Avet Pharmaceuticals Inc. The solution is preserved with phenol USP and pH adjusted as needed with sodium hydroxide NF/acetic acid USP. The medication must be kept protected from light and stored in the carton until use. It should be stored at a temperature between 20° to 25°C (68° to 77°F), with brief excursions permitted between 15° to 30°C (59° to 86°F). See package insert for directions for use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.