Product Images Bepreve
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Bepreve NDC 24208-629 by Bausch & Lomb Incorporated, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Carton 10mL

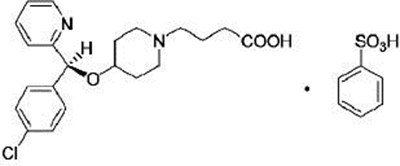
BEPREVE is a sterile ophthalmic solution containing bepotastine besilate used to treat eye allergies. It is distributed by Bausch Health US LLC and manufactured by Bausch & Lomb Incorporated in Tampa, FL under license from Senju Pharmaceutical Co, Ltd in Osaka, Japan. The product comes in a 10 mL container and is available in 1.5% and 15% concentrations. The usual dosage is one drop applied to each affected eye twice a day. The solution contains active and inactive ingredients with benzalkonium chloride as the preservative. It is important to instruct patients on precautions to avoid contamination. Store the unopened container at a temperature between 15 and 25 degrees Celsius.*
Carton 5mL

BEPREVE(bepotastine besilate ophthalmic solution) is a sterile 15% 5mL prescription medication used to treat eye allergies. Each mL contains 15 my of bepotastine besilate. To use, one drop should be instilled into the affected eye(s) twice a day. This medication is manufactured by and distributed by Bausch & Lomb, a division of Bausch Health US, LLC. It is sterile when manufactured and should be used only from the original unopened container to avoid contamination. It should be stored at 15°C to 25°C. No non-English characters found.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.