Product Images Triptodur
View Photos of Packaging, Labels & Appearance
Product Label Images
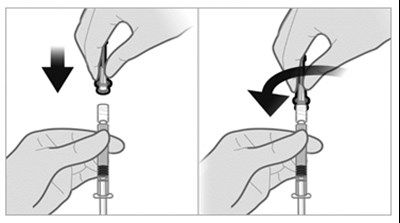
The following 12 images provide visual information about the product associated with Triptodur NDC 24338-150 by Azurity Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - Kit Carton - triptodur 12

Trlptodur is an injectable medication used for extended-release that contains triptorelin. It is administered intramuscularly as a single dose once every 24 weeks. The kit comes with one single-dose vial, one pre-filled syringe of diluent, and two thin-walled needles. This should be reconstituted with accompanying diluent before use. It should be stored at a controlled room temperature between 20 to 25°C, and it is not recommended to be frozen. The accompanying medication guide should be dispensed to each patient.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.