Product Images Dyanavel Xr
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 19 images provide visual information about the product associated with Dyanavel Xr NDC 24478-102 by Nextwave Pharmaceuticals, Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
16oz Label - dyanavel xr oral suspension and tablets 15

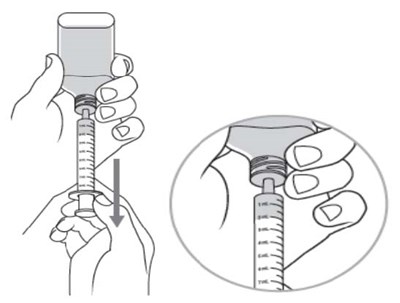
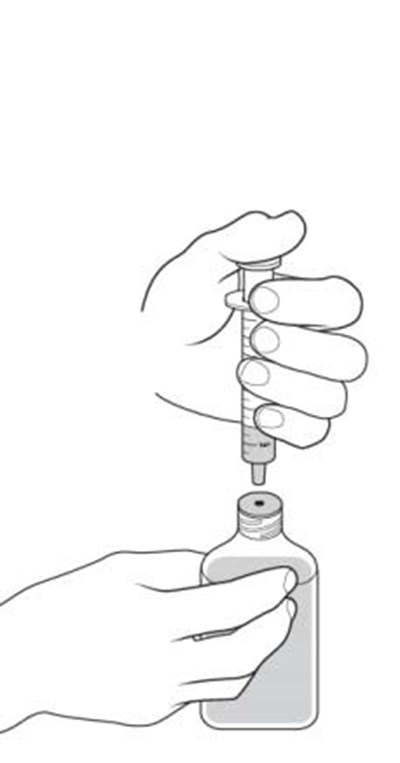
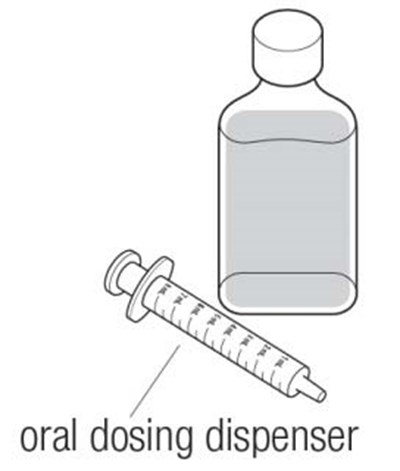
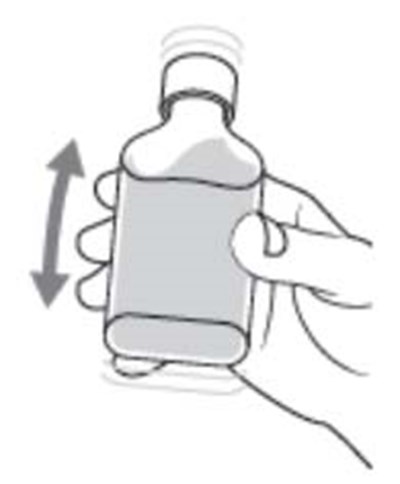
Dyanavel XR is an extended-release oral suspension containing amphetamine at a concentration of 2.5 mg/mL. The pharmacist should insert the bottle adapter firmly into the back of the bottle when dispensing the product. Patient instructions for use and medication guides should be provided with each dispensing. The product should be stored at room temperature. The product drains out when dispensing the last dose. GTIN code: 00324478102012.*
5mg Label - dyanavel xr oral suspension and tablets 16

This is a medication for Dyanavel XR, an extended-release Tablet that contains amphetamine base. The tablet can be chewed or swallowed whole. The medication guide should be given to each patient. It is recommended to follow the prescribing information for the full dosage recommendations. The medication comes in a USP container with a child-resistant closure and should be stored at room temperature. The warning advises keeping the medicine away from the reach of children. The product is manufactured by Tis Pharma, Inc. The remaining text contains product code information.*
10mg Label - dyanavel xr oral suspension and tablets 17

Each tablet contains 10 mg of amphetamine base and is intended for extended-release. The usual dosage information can be found in the prescription information. Dyanavel XR is a medication used to dispense amphetamines and comes in a USP container. The tablets can be chewed or swallowed. A warning is issued to keep medication away from children. The accompanying medication guide is necessary for each patient to receive. The manufacturer is Tris Pharma, Inc. and is only available by prescription with 30 tablets per container. The barcode at the end is a GTIN.*
15mg Label - dyanavel xr oral suspension and tablets 18

Each tablet contains 15 g of amphetamine base and is an extended-release tablet that can be swallowed whole or chewed. The usual dosage is not provided but the prescribing information for Dyanavel XR, which contains amphetamine, is mentioned. The tablets should be dispensed in a tight USP container with a child-resistant closure and kept out of reach of children. The manufacturer of the medication is Tis Pharma, and each patient is required to receive the accompanying Medication Guide. This text appears to be a label or prescription information for a medication and does not provide any additional information.*
20mg Label - dyanavel xr oral suspension and tablets 19

Dyanavel XR is a medication that comes in tablet form. Each tablet contains 20g of amphetamine sulfate. The recommended dosage information can be found on the prescribing information and should be dispensed in a tightly sealed USP container. The extended-release tablets are resistant to acid and can withstand temperature excursions between 15°C to 30°C. Each patient is required to receive a Medication Guide, and this medication should be kept out of the reach of children. Dyanavel XR is manufactured by Tris Pharma and is only available in packs of 30 tablets.*
Figure1 - dyanavel xr oral suspension and tablets 2

This is a report of plasma amphetamine levels in ng/mL for various drugs including Dyanavel XK 75, Dyanavel XR 7.5 mL, Mas Br. 30 mg, and Mas ER. 30 mg.*
Figure2 - dyanavel xr oral suspension and tablets 3

This appears to be the results of a drug test, specifically for amphetamines, showing different concentrations in ng/mL. The test was conducted using DYANAVEL XR in tablet and oral suspension form, with variations in how it was consumed (swallowed whole or chewed fast). The information provided could be useful for medical professionals, researchers, or individuals seeking to understand the effects of amphetamines on the body.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.