FDA Label for Prismasol Bgk2/3.5
View Indications, Usage & Precautions
- 1 INDICATIONS AND USAGE
- 2.1 ADMINISTRATION INSTRUCTIONS
- 2.2 DOSING CONSIDERATIONS
- 2.3 PREPARING THE SOLUTION
- OTHER
- 2.4 ADDING DRUGS TO THE SOLUTIONS
- 3 DOSAGE FORMS AND STRENGTHS
- 4 CONTRAINDICATIONS
- 5.1 ELECTROLYTE AND VOLUME ABNORMALITIES
- 5.2 HYPERGLYCEMIA WITH DEXTROSE CONTAINING SOLUTIONS
- 6 ADVERSE REACTIONS
- 7 DRUG INTERACTIONS
- 7.1 CITRATE
- 8.3 NURSING MOTHERS
- 8.4 PEDIATRIC USE
- 8.5 GERIATRIC USE
- 11 DESCRIPTION
- 12.1 MECHANISM OF ACTION
- 12.3 PHARMACOKINETICS
- 16 HOW SUPPLIED/STORAGE AND HANDLING
- STORAGE AND HANDLING
- PACKAGE/LABEL DISPLAY PANEL
Prismasol Bgk2/3.5 Product Label
The following document was submitted to the FDA by the labeler of this product Vantive Us Healthcare Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
1 Indications And Usage
PRISMASOL and PHOXILLUM solutions are indicated in pediatric and adult patients for use as a replacement solution in Continuous Renal Replacement Therapy (CRRT) to replace plasma volume removed by ultrafiltration and to correct electrolyte and acid-base imbalances. They may also be used in case of drug poisoning when CRRT is used to remove dialyzable substances.
2.1 Administration Instructions
Visually inspect PRISMASOL and PHOXILLUM for particulate matter and discoloration prior to administration.
Administration should only be under the direction of a physician competent in intensive care treatment including CRRT. Use only with extracorporeal dialysis equipment appropriate for CRRT.
The prepared solution is for single patient use only.
Aseptic technique should be used throughout administration to the patient.
Discard any unused solution.
2.2 Dosing Considerations
PRISMASOL replacement solutions contain 4 different combinations of active ingredients (8 different products with varying ingredient amounts). PHOXILLUM replacement solutions contain 2 different combinations of active ingredients (2 different products with varying ingredient amounts). PRISMASOL and PHOXILLUM are supplied in a two-compartment bag that must be mixed immediately prior to use [see Dosage and Administration (2.3)]:
• Small compartment A (250 mL) containing an electrolyte solution, and• Large compartment B (4750 mL) containing the buffer solution.• Before (pre-dilution) the hemofilter or hemodiafilter,• After (post-dilution) the hemofilter or hemodiafilter, or• Before and after the hemofilter or hemodiafilter.
See Table 1 for the concentrations of the active ingredients (after mixing) in these 10 different replacement solutions (total volume is 5 Liters).
| Ca2+ mEq/L | HCO3- mEq/L | K+ mEq/L | Mg2+ mEq/L | Na+ mEq/L | HPO42- mmol/L | Cl- mEq/L | Lactate mEq/L | Dextrose mg/dL | Osmolarity mOsm/L | |
|---|---|---|---|---|---|---|---|---|---|---|
| Ca2+ = calcium, HCO3- = bicarbonate, K+ = potassium, Mg2+ = magnesium, Na+ = sodium, HPO42- = phosphate, Cl- = chloride; osmolarity is estimated | ||||||||||
PRISMASOL Replacement Solutions | ||||||||||
BGK0/2.5 | 2.5 | 32 | 0 | 1.5 | 140 | 0 | 109 | 3 | 100 | 292 |
BGK4/2.5 | 2.5 | 32 | 4 | 1.5 | 140 | 0 | 113 | 3 | 100 | 300 |
BGK4/3.5 | 3.5 | 32 | 4 | 1 | 140 | 0 | 113.5 | 3 | 100 | 300 |
BGK2/3.5 | 3.5 | 32 | 2 | 1 | 140 | 0 | 111.5 | 3 | 100 | 296 |
BGK2/0 | 0 | 32 | 2 | 1 | 140 | 0 | 108 | 3 | 100 | 291 |
B22GK4/0 | 0 | 22 | 4 | 1.5 | 140 | 0 | 120.5 | 3 | 100 | 296 |
BGK4/0/1.2 | 0 | 32 | 4 | 1.2 | 140 | 0 | 110.2 | 3 | 100 | 295 |
BK0/0/1.2 | 0 | 32 | 0 | 1.2 | 140 | 0 | 106.2 | 3 | 0 | 282 |
PHOXILLUM Replacement Solutions | ||||||||||
BK4/2.5 | 2.5 | 32 | 4 | 1.5 | 140 | 1 | 114.5 | 0 | 0 | 294 |
B22K4/0 | 0 | 22 | 4 | 1.5 | 140 | 1 | 122 | 0 | 0 | 290 |
The mode of therapy, solute formulation, flow rates, and length of PRISMASOL and PHOXILLUM replacement therapy in CRRT should be established by a physician based on the patient’s clinical condition, blood concentration of phosphate and other electrolytes, acid-base and glucose balance. Administer either PRISMASOL or PHOXILLUM into the extracorporeal circuit:
2.3 Preparing The Solution
Use only if the overwrap is not damaged, all seals are intact, frangible pin or peel seal is not broken, and the solution is clear.
The solution may be warmed to 37°C/98.6°F inside of the overwrap to enhance patient comfort. However, only dry heat should be used. Solutions should not be heated in water or in a microwave oven. After heating, verify that the solution remains clear and contains no particulate matter.
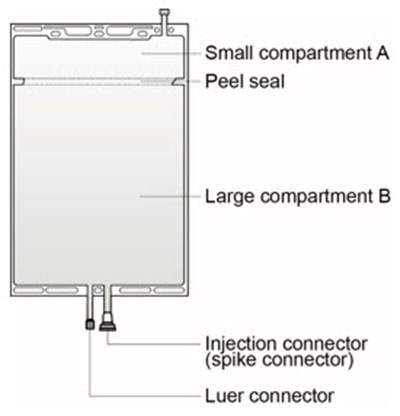
The solutions are supplied in two different two-compartment bags, one made of polyvinyl chloride with a frangible pin separating compartment A and B (see Figure 1) and one made of polyolefin with a peel seal separating compartment A and B (see Figure 6).
Follow the instructions below when connecting the solution bags for correct use of the access ports.
Other
Instructions for preparing solutions supplied in a two-compartment, polyvinyl chloride (PVC) bag with a red frangible pin:
Figure 1
Figure 2 | Step 1 Immediately before use, remove the overwrap from the bag and mix the solutions in the two different compartments. After removing the overwrap, inspect the container for leakage by pressing firmly on the bag. Discard the bag if any leakage is detected since sterility cannot be assured. As soon as the overwrap is removed, the reconstitution of compartments A and B should be done and the mixed solution should be used immediately. |
Figure 3 | Step 2 Make sure all the fluid from the small compartment A is transferred into the large compartment B. (See Figure 3 beside) |
Step 3 Rinse the small compartment A twice by pressing the mixed solution back into the small compartment A and then back into the large compartment B. (See Figure 3 beside) | |
Figure 4 | Step 4 When the small compartment A is empty, shake the large compartment B so that the contents mix completely. (See Figure 4 beside) |
Figure 5a | Step 5 The replacement line may be connected to the bag through either the luer connector or the injection connector (spike connector). |
Figure 5b | Step 5b If the injection connector (spike connector) is used, first remove the snap-off cap. Then introduce the replacement line spike through the rubber septum of the bag connector. (See Figure 5b beside) |
Instructions for preparing solutions supplied in a two-compartment, polyolefin bag with a peel seal:
Figure 6 |
Figure 7 | Step 1 Immediately before use, remove the overwrap from the bag and mix the solutions in the two different compartments. After removing the overwrap, inspect the container for leakage by pressing firmly on the bag. Discard the bag if any leakage is detected since sterility cannot be assured. As soon as the overwrap is removed, the reconstitution of compartments A and B should be done and the mixed solution should be used immediately. |
Figure 8 | Step 2 Squeeze with both hands on the large compartment until the peel seal between the two compartments is entirely open. Shake gently to mix. (See Figure 8 beside) |
The solution is now ready to use and the bag can be hung on the equipment. | |
Figure 9a | Step 3 The replacement line may be connected to the bag through either of the luer connector or the injection connector (spike connector). |
Step 3a The luer connector is a needle-less and swabbable connector. Remove the cap with a twist and pull motion, and connect the male luer lock on the replacement line to the female luer receptor on the bag. (See Figure 9a beside) | |
Ensure that the connection is fully seated and tighten. The connector is now open. Verify that the fluid is flowing freely during use. | |
When the replacement line is disconnected from the luer connector, the connector will close and the flow of the solution will stop. | |
Figure 9b | Step 3b If the injection connector (spike connector) is used, first remove the snap-off cap. Then introduce the replacement line spike through the rubber septum of the bag connector. (See Figure 9b beside) |
Ensure that the spike is fully inserted and verify that the fluid is flowing freely during use. |
PRISMASOL Solutions:
Phosphate: Phosphate up to 1.2 mmol/L may be added to the solution. If potassium phosphate is added, the total potassium concentration should not exceed 4 mEq/L.
PHOXILLUM Solutions:
Phosphate: Phosphate up to 0.2 mmol/L may be added to the solution. Use sodium phosphate if adding phosphate to bag. The total phosphate concentration should not exceed 1.2 mmol/L.
Pregnancy Category C
Animal reproduction studies have not been conducted with PRISMASOL and PHOXILLUM solutions. While there are no adequate and well controlled studies in pregnant women, appropriate administration of PRISMASOL and PHOXILLUM solutions with monitoring of fluid, electrolyte, acid-base and glucose balance, is not expected to cause fetal harm, or affect reproductive capacity. Maintenance of normal acid-base balance is important for fetal well-being.
2.4 Adding Drugs To The Solutions
After mixing, additional drugs may be added to the bag via injection connector (spike connector) in large compartment B. In general, drugs other than phosphate should be administered through a different access line.
When introducing drugs, use aseptic techniques and mix thoroughly. Do not use if there is a color change and/or the appearance of precipitates, insoluble complexes or crystals after addition of medication.
3 Dosage Forms And Strengths
See Table 1 for the concentrations of the active ingredients (after mixing) in these 10 different replacement solutions [see Dosage and Administration (2.2)].
4 Contraindications
PHOXILLUM and PRISMASOL replacement solutions are contraindicated in patients with known hypersensitivities to these products.
5.1 Electrolyte And Volume Abnormalities
PHOXILLUM and PRISMASOL solutions can affect electrolytes and volume and may result in hyperkalemia or hyperphosphatemia. Monitor hemodynamic status and fluid inputs and outputs, potassium, phosphorous, other electrolytes and acid-base balance throughout the procedure. Abnormalities may be corrected by changing the formulation of replacement solution and/or dialysate, supplementation, or adjusting flow rates appropriately [see Dosage and Administration (2)].
PHOXILLUM replacement solutions contain hydrogen phosphate, a weak acid that may increase the risk of metabolic acidosis.
5.2 Hyperglycemia With Dextrose Containing Solutions
The use of PRISMASOL replacement solutions containing dextrose may increase the risk for hyperglycemia in patients with impaired glucose tolerance. Patients may require initiation of or modification of antidiabetic therapy during treatment with PRISMASOL solutions containing dextrose. Monitor blood glucose.
6 Adverse Reactions
The following adverse reactions have been identified with other similar products and therefore, may occur with use of these products:
• Metabolic acidosis• Hypotension• Acid-base disorders• Electrolyte imbalance• Hyperphosphatemia (for phosphate containing solutions)• Fluid imbalance
7 Drug Interactions
As with the use of other replacement solutions, blood concentrations of dialyzable drugs may be reduced by CRRT due to their removal by the hemofilter or hemodiafilter. The blood concentrations of certain drugs may need to be monitored and appropriate therapy implemented to correct for removal during treatment.
7.1 Citrate
When used as an anticoagulant, citrate contributes to the overall buffer load and can reduce plasma calcium levels. Select the PRISMASOL/PHOXILLUM formulation(s) accordingly.
8.3 Nursing Mothers
The components of PRISMASOL and PHOXILLUM solutions are excreted in human milk. Appropriate administration of PRISMASOL and PHOXILLUM solutions with monitoring of fluid, electrolyte, acid-base and glucose balance, is not expected to harm a nursing infant.
8.4 Pediatric Use
Safety and effectiveness have been established based on published clinical data of CRRT replacement solutions with compositions similar to PRISMASOL and PHOXILLUM used in adults and two hemofiltration studies in pediatric patients, including a study of newborns to 17 years old.
8.5 Geriatric Use
The experience with PRISMASOL and PHOXILLUM solutions in geriatric patients has not identified novel concerns.
11 Description
PRISMASOL and PHOXILLUM solutions are clear, sterile, free of bacterial endotoxins and contain no bacteriostatic or antimicrobial agents. These solutions are used in Continuous Renal Replacement Therapies (CRRT) as a replacement solution in hemofiltration and hemodiafiltration. Depending on the product (see Table 2), the two compartments contain:
Calcium chloride, USP, is chemically designated calcium chloride dihydrate (CaCl2 • 2H2O).
Magnesium chloride, USP, is chemically designated magnesium chloride hexahydrate (MgCl2 • 6H2O).
Sodium chloride, USP, is chemically designated NaCl.
Potassium chloride, USP, is chemically designated KCl.
Sodium bicarbonate, USP, is chemically designated NaHCO3.
Dextrose, USP, is chemically designated D-Glucose anhydrous (C6H12O6) or D-Glucose monohydrate (C6H12O6 • H2O).
Lactic acid, USP, is chemically designated CH3CH(OH)COOH.
Dibasic sodium phosphate, USP, is chemically designated as disodium hydrogen phosphate, dihydrate (Na2HPO4 • 2H2O)
| Compartment A (g/L) | Compartment B (g/L) | |||||||
|---|---|---|---|---|---|---|---|---|
| Calcium Chloride ∙ 2H2O | Magnesium Chloride ∙ 6H2O | Dextrose anhydrous (as monohydrate) | Lactic Acid | Sodium Chloride | Sodium bicarbonate | Potassium Chloride | Sodium Phosphate ∙ 2H2O | |
PRISMASOL SOLUTIONS | ||||||||
BGK 0/2.5 | 3.68 | 3.05 | 20 (22) | 5.40 | 6.46 | 3.09 | 0 | 0 |
BGK 4/2.5 | 3.68 | 3.05 | 20 (22) | 5.40 | 6.46 | 3.09 | 0.314 | 0 |
BGK 4/3.5 | 5.15 | 2.03 | 20 (22) | 5.40 | 6.46 | 3.09 | 0.314 | 0 |
BGK 2/3.5 | 5.15 | 2.03 | 20 (22) | 5.40 | 6.46 | 3.09 | 0.157 | 0 |
BGK 2/0 | 0 | 2.03 | 20 (22) | 5.40 | 6.46 | 3.09 | 0.157 | 0 |
B22GK 4/0 | 0 | 3.05 | 20 (22) | 5.40 | 7.07 | 2.21 | 0.314 | 0 |
BK 0/0/1.2 | 0 | 2.44 | 0 (0) | 5.40 | 6.46 | 3.09 | 0 | 0 |
BGK 4/0/1.2 | 0 | 2.44 | 20 (22) | 5.40 | 6.46 | 3.09 | 0.314 | 0 |
PHOXILLUM SOLUTIONS | ||||||||
BK 4/2.5 | 3.68 | 3.05 | 0 (0) | 0 | 6.34 | 3.09 | 0.314 | 0.187 |
B22K 4/0 | 0 | 3.05 | 0 (0) | 0 | 6.95 | 2.21 | 0.314 | 0.187 |
The pH of the final solution is in the range of 7.0 to 8.5.
12.1 Mechanism Of Action
PRISMASOL and PHOXILLUM solutions are pharmacologically inactive. The electrolyte concentrations in the solutions are chosen to restore plasma levels to clinically desired concentrations or maintain plasma levels at the desired concentrations.
PRISMASOL and PHOXILLUM solutions are used as replacement solution to replace water and electrolytes removed during hemofiltration and hemodiafiltration. Bicarbonate (or precursor lactate) in the solution is used as an alkalinizing buffer to restore acid-base balance to a clinically desirable level.
12.3 Pharmacokinetics
The distribution of electrolytes, bicarbonate, and dextrose is determined by the patient's clinical condition, metabolic status, and residual renal function.
The elimination and replacement of water, electrolytes and buffer depend on the patient's electrolyte and acid-base balance, metabolic status, residual renal function and ongoing physiologic losses through intestinal, respiratory and cutaneous routes.
16 How Supplied/Storage And Handling
PRISMASOL and PHOXILLUM solutions are supplied in a two-compartment bag made of either polyvinyl chloride (PVC) or polyolefin. The 5000 mL bag is composed of a small compartment (250 mL) and a large compartment (4750 mL). The two compartments are separated by a red frangible pin in the PVC bag and by a peel seal in the polyolefin bag.
The bag is overwrapped with a transparent overwrap. See Table 2 for the concentrations of the active ingredients in each compartment for each product [see Description (11)].
Container | Fill Volume | NDC | Bag Type | |
PRISMASOL Solutions | ||||
PRISMASOL BGK0/2.5 | 5000 mL | 24571-108-05 | PVC | |
PRISMASOL BGK4/2.5 | 5000 mL | 24571-105-05 | ||
PRISMASOL BGK4/3.5 | 5000 mL | 24571-104-05 | ||
PRISMASOL BGK2/3.5 | 5000 mL | 24571-103-05 | ||
PRISMASOL BGK2/0 | 5000 mL | 24571-102-05 | ||
PRISMASOL B22GK4/0 | 5000 mL | 24571-111-05 | ||
PRISMASOL BK0/0/1.2 | 5000 mL | 24571-113-05 | ||
PRISMASOL BGK4/0/1.2 | 5000 mL | 24571-114-05 | ||
PRISMASOL BGK0/2.5 | 5000 mL | 24571-108-06 | Polyolefin | |
PRISMASOL BGK4/2.5 | 5000 mL | 24571-105-06 | ||
PRISMASOL BGK2/3.5 | 5000 mL | 24571-103-06 | ||
PRISMASOL BGK2/0 | 5000 mL | 24571-102-06 | ||
PRISMASOL B22GK4/0 | 5000 mL | 24571-111-06 | ||
PRISMASOL BK0/0/1.2 | 5000 mL | 24571-113-06 | ||
PRISMASOL BGK4/0/1.2 | 5000 mL | 24571-114-06 | ||
PHOXILLUM Solutions | ||||
PHOXILLUM BK4/2.5 | 5000 mL | 24571-116-05 | PVC | |
PHOXILLUM B22K4/0 | 5000 mL | 24571-117-05 | ||
PHOXILLUM BK4/2.5 | 5000 mL | 24571-116-06 | Polyolefin | |
PHOXILLUM B22K4/0 | 5000 mL | 24571-117-06 | ||
Not all formulations may be marketed.
Storage And Handling
Storage conditions
Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). [See USP
Controlled Room Temperature]
Do not freeze or expose to excessive heat. Do not use if precipitate has formed or if container seals have
been damaged.
Manufactured for:
Baxter Healthcare Corporation
One Baxter Parkway
Deerfield, Illinois 60015
Baxter, Gambro, Phoxillum and PrismaSol are trademarks of Baxter International Inc., or its subsidiaries
Package/Label Display Panel
! Symbol MIX Compartments ! Symbol
D14000292 Ver. 1.0
Mixing steps:
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Bar Code
NDC# 24571-108-06
0K+
mEq/L
2.5 Ca2+
mEq/L
Rx only
PrismaSol® BGK0/2.5
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Calcium chloride • 2H2O | 3.68 | |
Magnesium chloride • 6H2O | 3.05 | |
Dextrose anhydrous | 20.0 | |
(as dextrose monohydrate) | 22.0 | |
Sodium chloride | 6.46 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 3.09 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
A
250 mL
B
4750 mL
After reconstitution, A + B | ||||||||
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 1.25 | 0.75 | 140 | 109.0 | 3.0 | 32 | 0 | 5.5 |
mEq/L | 2.5 | 1.5 | 140 | 109.0 | 3.0 | 32 | 0 | (100 mg/dL) |
| ||||||||
Mix both compartments before use. | Do not freeze or expose to excessive heat. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414091613 Product No: 110240
Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000293 2015-10
PrismaSol BGK0/2.5
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 110240 S
NDC# 24571-108-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000293 Rev. 2016-04
! Symbol MIX Compartments ! Symbol
D14000292 Ver. 1.0
Mixing steps:
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Bar Code
NDC# 24571-105-06
4K+
mEq/L
2.5 Ca2+
mEq/L
Rx only
PrismaSol® BGK4/2.5
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Calcium chloride • 2H2O | 3.68 | |
Magnesium chloride • 6H2O | 3.05 | |
Dextrose anhydrous | 20.0 | |
(as dextrose monohydrate) | 22.0 | |
Sodium chloride | 6.46 | |
Potassium chloride | 0.314 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 3.09 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
A
250 mL
B
4750 mL
After reconstitution, A + B | ||||||||
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 1.25 | 0.75 | 140 | 113.0 | 3.0 | 32 | 4.0 | 5.5 |
mEq/L | 2.5 | 1.5 | 140 | 113.0 | 3.0 | 32 | 4.0 | (100 mg/dL) |
| ||||||||
Mix both compartments before use. | Do not freeze or expose to excessive heat. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414091637 Product No: 110242
Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000295 2015-10
PrismaSol BGK4/2.5
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 110242 S
NDC# 24571-105-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000295 Rev. 2016-04
! Symbol BREAK red pin and MIX
compartment A with compartment B Compartment A Symbol
D14000299 Rev. 2015-09
Break red pin and mix. ↑ Symbol Compartment B Symbol
4K+
mEq/L
3.5 Ca2+
mEq/L
PrismaSol® BGK4/3.5
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Calcium chloride • 2H2O | 5.15 | |
Magnesium chloride • 6H2O | 2.03 | |
Dextrose anhydrous | 20.0 | |
(as dextrose monohydrate) | 22.0 | |
Sodium chloride | 6.46 | |
Potassium chloride | 0.314 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 3.09 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
Rx only
A
250 mL
B
4750 mL
After reconstitution, A + B
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 1.75 | 0.5 | 140 | 113.5 | 3.0 | 32 | 4.0 | 5.5 |
mEq/L | 3.5 | 1.0 | 140 | 113.5 | 3.0 | 32 | 4.0 | (100 mg/dL) |
Mix both compartments before use. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414120160 Product# 115633
Batch No. and expiry date are printed on the
back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000306 Rev. 2015-09
PrismaSol BGK4/3.5
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 1105633
NDC# 24571-104-05
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000306 Rev. 2015-09
! Symbol MIX Compartments ! Symbol
D14000292 Ver. 1.0
Mixing steps:
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Bar Code
NDC# 24571-103-06
2K+
mEq/L
3.5 Ca2+
mEq/L
Rx only
PrismaSol® BGK2/3.5
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Calcium chloride • 2H2O | 5.15 | |
Magnesium chloride • 6H2O | 2.03 | |
Dextrose anhydrous | 20.0 | |
(as dextrose monohydrate) | 22.0 | |
Sodium chloride | 6.46 | |
Potassium chloride | 0.157 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 3.09 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
A
250 mL
B
4750 mL
After reconstitution, A + B | ||||||||
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 1.75 | 0.5 | 140 | 111.5 | 3.0 | 32 | 2.0 | 5.5 |
mEq/L | 3.5 | 1.0 | 140 | 111.5 | 3.0 | 32 | 2.0 | (100 mg/dL) |
Mix both compartments before use. | Do not freeze or expose to excessive heat. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414091644 Product No: 110243
Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000296 2015-10
PrismaSol BGK2/3.5
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 110243 S
NDC# 24571-103-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000296 Rev. 2016-04
! Symbol MIX Compartments ! Symbol
D14000292 Ver. 1.0
Mixing steps:
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Bar Code
NDC# 24571-102-06
2K+
mEq/L
0Ca2+
mEq/L
Rx only
PrismaSol® BGK4/3.5
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Magnesium chloride • 6H2O | 2.03 | |
Dextrose anhydrous | 20.0 | |
(as dextrose monohydrate) | 22.0 | |
Sodium chloride | 6.46 | |
Potassium chloride | 0.157 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 3.09 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
Rx only
A
250 mL
B
4750 mL
After reconstitution, A + B | ||||||||
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 0 | 0.5 | 140 | 108.0 | 3.0 | 32 | 2.0 | 5.5 |
mEq/L | 0 | 1.0 | 140 | 108.0 | 3.0 | 32 | 2.0 | (100 mg/dL) |
Mix both compartments before use. | Do not freeze or expose to excessive heat. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414091651 Product No: 110244
Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000297 2015-10
PrismaSol BGK2/0
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 110244 S
NDC# 24571-102-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000297 Rev. 2016-04
! Symbol MIX Compartments ! Symbol
D14000292 Ver. 1.0
Mixing steps:
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Bar Code
NDC# 24571-111-06
4K+
mEq/L
Bicarbonate 22
0Ca2+
mEq/L
Rx only
PrismaSol®B22GK4/0
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Magnesium chloride • 6H2O | 3.05 | |
Dextrose anhydrous | 20.0 | |
(as dextrose monohydrate) | 22.0 | |
Sodium chloride | 7.07 | |
Potassium chloride | 0.314 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 2.21 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
A
250 mL
B
4750 mL
After reconstitution, A + B | ||||||||
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 0 | 0.75 | 140 | 120.5 | 3.0 | 22 | 4.0 | 5.5 |
mEq/L | 0 | 1.5 | 140 | 120.5 | 3.0 | 22 | 4.0 | (100 mg/dL) |
Mix both compartments before use. | Do not freeze or expose to excessive heat. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414116781 Product No: 115001
Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000298 2015-10
PrismaSol B22GK4/0
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 115001 S
NDC# 24571-111-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000298 Rev. 2016-04
! Symbol MIX Compartments ! Symbol
D14000292 Ver. 1.0
Mixing steps:
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Bar Code
NDC# 24571-113-06
0K+
mEq/L
0Ca2+
mEq/L
Rx only
PrismaSol® BK0/0/1.2
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Magnesium chloride • 6H2O | 2.44 | |
Sodium chloride | 6.46 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 3.09 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
A
250 mL
B
4750 mL
After reconstitution, A + B | ||||||||
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 0 | 0.6 | 140 | 106.2 | 3.0 | 32 | 0 | 0 |
mEq/L | 0 | 1.2 | 140 | 106.2 | 3.0 | 32 | 0 | (0 mg/dL) |
Mix both compartments before use. | Do not freeze or expose to excessive heat. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414091309 Product No: 110239
Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000292 2015-09
PrismaSol BK0/0/1.2
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 110239 S
NDC# 24571-113-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000292 Rev. 2016-04
! Symbol MIX Compartments ! Symbol
D14000292 Ver. 1.0
Mixing steps:
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Bar Code
NDC# 24571-114-06
4K+
mEq/L
0Ca2+
mEq/L
Rx only
PrismaSol® BGK4/0/1.2
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution each 1000 mL contains (g) | A | B |
Magnesium chloride • 6H2O | 2.44 | |
Dextrose anhydrous | 20.0 | |
(as dextrose monohydrate) | 22.0 | |
Sodium chloride | 6.46 | |
Potassium chloride | 0.314 | |
Lactic acid | 5.40 | |
Sodium bicarbonate | 3.09 | |
Water for injections q.s, Carbon dioxide for pH adjustment | ||
A
250 mL
B
4750 mL
After reconstitution, A + B | ||||||||
Calcium | Magnesium | Sodium | Chloride | Lactate | Bicarbonate | Potassium | Dextrose | |
mmol/L | 0 | 0.6 | 140 | 110.2 | 3.0 | 32 | 4.0 | 5.5 |
mEq/L | 0 | 1.2 | 140 | 110.2 | 3.0 | 32 | 4.0 | (100 mg/dL) |
Mix both compartments before use. | Do not freeze or expose to excessive heat. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414091620 Product No: 110241
Batch No. and expiry date are printed on the back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000294 2015-10
PrismaSol BK4/0/1.2
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 110241 S
NDC# 24571-114-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000294 Rev. 2016-04
! Symbol MIX compartment A with compartment B Compartment A Symbol
D14000349 Ver. 1.0
Mixing steps
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Compartment B Symbol
Bar Code
NDC# 24571-116-06
4 K+
mEq/L
1 Phosphate
mmol/L
2.5 Ca2+
mEq/L
Phoxillum BK4/2.5
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution, each 1000 mL contains (g): | A | B |
Calcium chloride • 2H2O | 3.68 | |
Magnesium chloride • 6H2O | 3.05 | |
Sodium chloride | 6.34 | |
Potassium chloride | 0.314 | |
Sodium bicarbonate | 3.09 | |
Dibasic sodium phosphate • 2H2O | 0.187 | |
Water for injections q.s | ||
Rx only
A
250 mL
B
4750 mL
After reconstitution, A + B
Calcium | Magnesium | Sodium | Chloride | Bicarbonate | Potassium | Phosphate HPO42− | Dextrose | |
mmol/L | 1.25 | 0.75 | 140 | 114.5 | 32 | 4.0 | 1 | 0 |
mEq/L | 2.5 | 1.5 | 140 | 114.5 | 32 | 4.0 | (1 mmol/L) | (0 mg/dL) |
Mix both compartments before use. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414116040 Product# 114905
Batch No. and expiry date are printed on the
back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000349 Ver. 1.0
Phoxillum BK4/2.5
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 114905
NDC 24571-116-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000349 Ver. 1.0
! Symbol MIXcompartment A with compartment B Compartment A Symbol
D14000349 Ver. 1.0
Mixing steps
1 SQUEEZE TOP corners to break seal
2 Squeeze BAG SIDES to fully open seal
Compartment B Symbol
Bar Code
NDC# 24571-117-06
4 K+
mEq/L
0 Ca2+
mEq/L
22 Bicarbonate
mEq/L
1 Phosphate
mmol/L
Phoxillum B22K4/0
Replacement Solution for Continuous Renal Replacement Therapy
Before reconstitution, each 1000 mL contains (g): | A | B |
Magnesium chloride • 6H2O | 3.05 | |
Sodium chloride | 6.95 | |
Potassium chloride | 0.314 | |
Sodium bicarbonate | 2.21 | |
Dibasic sodium phosphate • 2H2O | 0.187 | |
Water for injections q.s | ||
Rx only
A
250 mL
B
4750 mL
After reconstitution, A + B
Calcium | Magnesium | Sodium | Chloride | Bicarbonate | Potassium | Phosphate | Dextrose | |
mmol/L | 0 | 0.75 | 140 | 122.0 | 22 | 4.0 | 1 | 0 |
mEq/L | 0 | 1.5 | 140 | 122.0 | 22 | 4.0 | (1 mmol/L) | (0 mg/dL) |
Mix both compartments before use. |
5000 mL
Gambro Logo
GAMBRO®
EAN-14: 07332414116057 Product# 114906
Batch No. and expiry date are printed on the
back of the bag.
Manufactured for:
Baxter Healthcare Corporation
Deerfield IL 60015 USA
Made in Italy
R E P L A C E M E N T
Solution for Continuous Renal Replacement Therapy
D11000350 Ver. 1.0
Phoxillum B22K4/0
Replacement Solution for Continuous Renal Replacement Therapy
REF Cat. # 114906
NDC 24571-117-06
LOT # Expiry
Rx Only
2 x 5000 mL
Store at +20°C top +25°C (+68°F to +77°F); excursions permitted to +15°C to +30°C (+59°F to +86°F).
[See USP Controlled Temperature]. Do not freeze or expose to excessive heat.
D12000350 Ver. 1.0
* Please review the disclaimer below.