FDA Label for Derma-glove
View Indications, Usage & Precautions
Derma-glove Product Label
The following document was submitted to the FDA by the labeler of this product Rwm Technologies. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Description
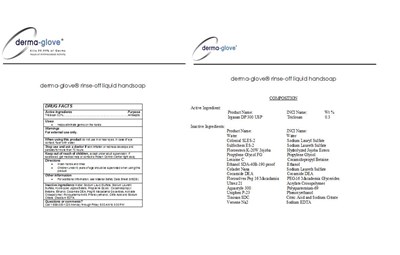
DERMA-GLOVE(R) rinse-off liquid handsoap
Otc - Active Ingredient
Active Ingredient
Triclosan 0.3%.............................................Antiseptic
Otc - Purpose
Antiseptic hand soap
Indications & Usage
- Helps eliminate germs on the hands
Warnings And Precautions
Warnings
For external use only.
Otc - When Using
When using this product do not use in or near eyes. In case of eye contact, flush with water.
Otc - Stop Use
Stop use and ask a doctor if skin irritation or redness develops and persists for more than 72 hours.
Otc - Keep Out Of Reach Of Children
Keep out of the Reach of Children, except under adult supervision. If swallowed, get medical help or contact a Poison Control Center right away.
Dosage & Administration
Directions:
- Wash hands and rinse
- Children under 6 years of age should be supervised when using this product
General Precautions
For additional information, see Material Safety Data Sheet (MSDS).
Inactive Ingredient
Water, Sodium Lauryl Sulfate, Sodium Laureth Sulfate, Hydrolyzed Jojoba Esters, Propylene Glycol, Cocamidopropyl Betaine, Ethanol, Cocamide DEA, Peg16 Macadamia Glycerides, Acrylate Crosspolymer, Polyquaternium-69, Phenoxyethanol, Citric Acid and Sodium Citrate, Disodium EDTA
Otc - Questions
Questions or Comments?
Call 1-888-300-1225 Monday through Friday 8:00 AM to 5:00 PM
Package Label.Principal Display Panel
LABEL
* Please review the disclaimer below.