Product Images Buspirone Hydrochloride
View Photos of Packaging, Labels & Appearance
- b7db4c3d 72e4 420c 9b81 119646c13c85 01
- b7db4c3d 72e4 420c 9b81 119646c13c85 02
- b7db4c3d 72e4 420c 9b81 119646c13c85 03
- b7db4c3d 72e4 420c 9b81 119646c13c85 04
- b7db4c3d 72e4 420c 9b81 119646c13c85 05
- b7db4c3d 72e4 420c 9b81 119646c13c85 06
- b7db4c3d 72e4 420c 9b81 119646c13c85 07
- b7db4c3d 72e4 420c 9b81 119646c13c85 08
- b7db4c3d 72e4 420c 9b81 119646c13c85 09
- b7db4c3d 72e4 420c 9b81 119646c13c85 10
- b7db4c3d 72e4 420c 9b81 119646c13c85 11
- b7db4c3d 72e4 420c 9b81 119646c13c85 12
- b7db4c3d 72e4 420c 9b81 119646c13c85 13
- b7db4c3d 72e4 420c 9b81 119646c13c85 14
- b7db4c3d 72e4 420c 9b81 119646c13c85 15
- b7db4c3d 72e4 420c 9b81 119646c13c85 16
- b7db4c3d 72e4 420c 9b81 119646c13c85 17
- b7db4c3d 72e4 420c 9b81 119646c13c85 18
- b7db4c3d 72e4 420c 9b81 119646c13c85 19
- b7db4c3d 72e4 420c 9b81 119646c13c85 20
Product Label Images
The following 20 images provide visual information about the product associated with Buspirone Hydrochloride NDC 24689-785 by Apnar Pharma Lp, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
b7db4c3d 72e4 420c 9b81 119646c13c85 12

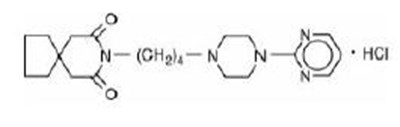
This is a description of a medication with the active ingredient Buspirone Hydrochloride in tablet form. Each tablet contains 5 mg of buspirone hydrochloride. The usual dosage is provided in the package insert. Storage instructions recommend keeping it between 20°C to 25°C. The medication should be kept in a tight, light-resistant container with a child-resistant closure. The text also includes details such as NDC code, quantity of tablets (500 tablets), manufacturer (Inventia Healthcare Limited in India), and marketer information. All medications should be kept out of the reach of children.*
b7db4c3d 72e4 420c 9b81 119646c13c85 13

This text appears to be prescription information for medication containing Hydrochloride tablets. It includes the address, dosage information, storage instructions, and a note for the pharmacist to dispense with an attached patient information leaflet. The medication should be stored between 20-25°C (68-77°F) in a controlled room temperature. Additionally, it advises to keep the medication out of reach of children and mentions a code number. Please consult a medical professional for proper usage and information.*
b7db4c3d 72e4 420c 9b81 119646c13c85 14

This is a detailed medication label for Buspirone Hydrochloride Tablets, USP, containing 500 tablets with each tablet containing 10 mg of buspirone hydrochloride. The usual dosage information can be found in the package insert. It is recommended to store the tablets at 20° to 25°C. The medication should be kept in a tight, light-resistant container with a child-resistant closure. The manufacturer is Inventia Healthcare Limited in India, and the product is marketed by Apnar Pharma LP in the USA.*
b7db4c3d 72e4 420c 9b81 119646c13c85 15

This text appears to be a label for tablets, possibly for a medication containing hydrochloride. The text mentions the tablets meet USP standards, should be stored at controlled room temperature, are light resistant, and need to be kept out of reach of children. Additionally, there is a code number provided at the bottom of the label. The unvarnished area size is listed as 45 x 15 mm.*
b7db4c3d 72e4 420c 9b81 119646c13c85 16

This is a description of busPIRone Hydrochloride Tablets, USP with a strength of 30 mg per tablet. The packaging contains 60 tablets and the prescription should be dispensed with the attached patient information leaflet. The tablets are to be stored at 20° to 25°C (68° to 77°F) in a tight, light-resistant container. The medication is manufactured by Inventia Healthcare Limited in India and marketed by Apnar Pharma LP. The usual dosage information is available in the package insert. It is important to keep the medication out of reach of children. Manufacturer's address: Additional Ambernath W1.0.C., Ambernath (East) - 421506, INDIA.*
b7db4c3d 72e4 420c 9b81 119646c13c85 17

This is a description of a medication package containing 500 tablets of buspirone hydrochloride, each tablet containing 15 mg of the active ingredient. The package includes a patient information leaflet and should be stored at 20° to 25°C. The medication is to be dispensed in a tight, light-resistant container with a child-resistant closure. The manufacturing details indicate it was made by Inventia Healthcare Limited in India and marketed by Apnar Pharma LP in California. The total unvarnished area is mentioned as 5020 mm.*
b7db4c3d 72e4 420c 9b81 119646c13c85 20

This is a product description for buspirone hydrochloride tablets. Each tablet contains 15 mg of buspirone hydrochloride, USP. The usual dosage instructions can be found in the package insert. The tablets are packaged in a container holding 100 tablets. It is recommended to store the tablets at 20° to 25°C (68° to 77°F) and keep them away from children. The manufacturer is Inventia Healthcare Limited in India, and the marketing company is Apnar Pharma LP based in California, USA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.