Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- Donepezil Hydrochloride Tablets - done10mg30tab
- Donepezil Hydrochloride Tablets - done5mg30tab
- fig1 - donepezil fig1
- fig10 - donepezil fig10
- fig2 - donepezil fig2
- fig3 - donepezil fig3
- fig4 - donepezil fig4
- fig5 - donepezil fig5
- fig6 - donepezil fig6
- fig7 - donepezil fig7
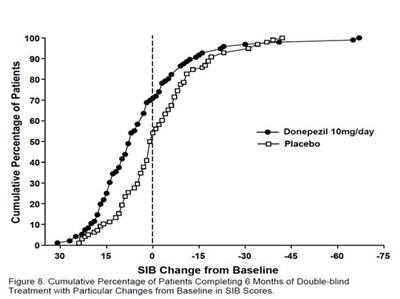
- fig8 - donepezil fig8
- fig9 - donepezil fig9
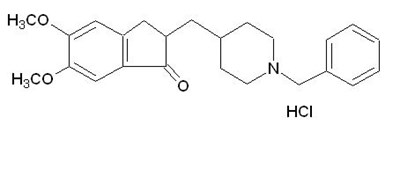
- structure - donepezil str
Product Label Images
The following 13 images provide visual information about the product associated with Donepezil Hydrochloride NDC 24724-029 by Indicus Pharma Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Donepezil Hydrochloride Tablets - done10mg30tab

This is a medication called Donepezil Hydrochloride Tablet that comes in a package of 30 tablets, each containing 10mg of the active ingredient. The LotNo. is also provided. The medication is only available with a prescription and should be kept at controlled room temperature. There is an accompanying patient information sheet, and the medication should be dispensed only by a pharmacist based on the prescription provided. The manufacturer of the medicine is listed as Indicus BHAMA IC, and it is produced in Daman, India.*
Donepezil Hydrochloride Tablets - done5mg30tab

This is a prescription-only medication consisting of tablets coated with a fim containing 5mg of the active ingredient donepezil hydrochloride. It should be stored at room temperature in tightly sealed containers and must be dispensed with an attached patient information sheet. Dosage and usage information is to be found in the accompanying prescribing information. The medication came from USV Limited with an address in India and has a lot number and expiration date. The NDC number is 24724-028-03 and the manufacturer is identified as M.*
fig1 - donepezil fig1

This is a graph that shows the change in ADAS-cog score for patients completing 24 weeks of treatment with donepezil 10mg/day, donepezil 5mg/day, or placebo. There are three lines, each representing one treatment group. The X-axis shows weeks of drug treatment, while the Y-axis shows the change from baseline in ADAS-cog score. The graph shows that patients treated with donepezil 10mg/day and donepezil 5mg/day had clinical improvement compared to placebo over the course of the 24 weeks.*
fig10 - donepezil fig10

The given text describes a figure labeled as "ADCS-ADL Change from Baseline". The figure illustrates the "Cumulative Percentage of Patients Completing 6 Months of Double-blind Treatment with Particular Changes from Baseline in ADCS-ADL-Severe Scores". The two treatments that were compared were donepezil 10 mg/day and placebo. However, there is not enough information available to provide any further context regarding the study.*
fig2 - donepezil fig2

The text describes a study on the use of Donepezil at different dosages of 10 mg/day and 5 mg/day. The study shows a figure indicating the percentage of patients who completed 24 weeks of double-blind treatment with specified changes from baseline ADAS-cog scores. The study had a placebo group and randomized patients. The percentage of patients who completed the study was 80% for placebo, 85% for 5 mg/day, and 68% for 10 mg/day.*
fig3 - donepezil fig3

The text describes a table or graph related to a study of the effectiveness of Donepezil. It includes dosages, a placebo, and scores measuring improvement or decline. The last line describes a frequency distribution related to the CIBIC-plus rating at 24 weeks.*
fig4 - donepezil fig4

This excerpt shows data related to the change in ADAS-cog score for patients completing a 15-week study while taking Donepezil 10 mg/day, Donepezil § mgday, or a placebo. The figure demonstrates the time course of the change from baseline in ADAS-cog score. No further information is available.*
fig5 - donepezil fig5

The text describes a clinical trial evaluating the effects of Donepezil on Alzheimer's patients. Three groups were assigned, with dosages of 0mg (placebo), 5mg/day, and 10mg/day. The "Change in ADAS-cog" scores were evaluated at baseline and throughout the study, and the "Cumulative Percentage of Patients" shows that patients who took Donepezil scored better than the placebo group, with the 10mg/day group achieving the best results. The trial was completed by 93% of the placebo group, 90% of the 5mg/day group, and 82% of the 10mg/day group.*
fig6 - donepezil fig6

This is a graph showing the frequency distribution of CIBIC plus scores at Week 12 for patients who were given Donepezil 10mg/day or 5mg/day compared to a placebo. The CIBIC-plus Rating measures the change in cognitive function in patients with Alzheimer's disease. There are four categories of improvement (Markedly, Moderately, Minimally, or No Change) and three categories of worsening (Minimally, Moderately, or Markedly). The x-axis shows the categories of improvement or worsening, while the y-axis shows the percentage of patients falling into each category.*
fig7 - donepezil fig7

This is a graph showing the time course of change from baseline in SIB score for patients who completed 6 months of treatment either with Donepezil 10 mg/day or with a placebo. The baseline mean score and standard error are also provided.*
fig9 - donepezil fig9

This is a graph that shows the improvement and decline in patients who completed 6 months of treatment with either a placebo or donepezil 10mg/day for Alzheimer's disease, as measured by the ADCS-ADL-Severe Score. The x-axis indicates the time course, while the y-axis indicates the change from baseline. The graph shows that patients treated with donepezil had a notable improvement compared to patients treated with a placebo.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.