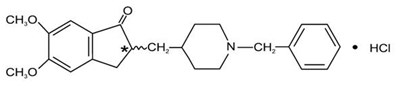
Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- 01 - 30406488 figure 01
- figure2 - 30406488 figure 02
- figure-03 - 30406488 figure 03
- figure-04 - 30406488 figure 04
- figure-05 - 30406488 figure 05
- figure-06 - 30406488 figure 06
- figure-07 - 30406488 figure 07
- 08 - 30406488 figure 08
- 09 - 30406488 figure 09
- 10 - 30406488 figure 10
- 11 - 30406488 figure 11
- 12 - 30406488 figure 12
- 13 - 30406488 figure 13
- 14 - 30406488 figure 14
- 15 - 30406488 figure 15
- 16 - 30406488 figure 16
- 17 - 30406488 figure 17
Product Label Images
The following 17 images provide visual information about the product associated with Donepezil Hydrochloride NDC 24979-004 by Upsher-smith Laboratories, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
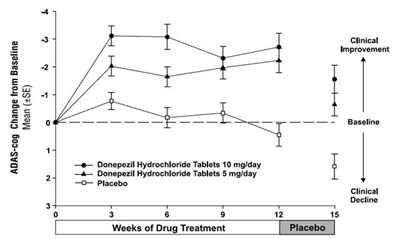
figure2 - 30406488 figure 02
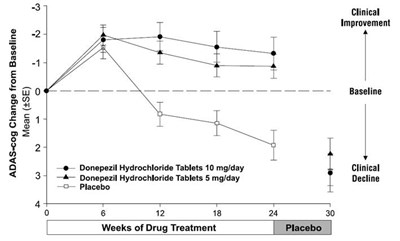
This is a graph showing the change from baseline in ADAS-cog scores over 24 weeks of drug treatment for donepezil hydrochloride tablets at 5mg and 10mg per day and placebo. The graph indicates weeks along the x-axis and mean ADAS-cog scores along the y-axis. There are two lines, one showing the ADAS-cog scores for donepezil hydrochloride tablets at 5mg per day and the other for donepezil hydrochloride tablets at 10 mg per day. The third line represents the placebo. Clinical improvement is represented by decreasing ADAS-cog scores and clinical decline is represented by increasing ADAS-cog scores.*
figure-03 - 30406488 figure 03
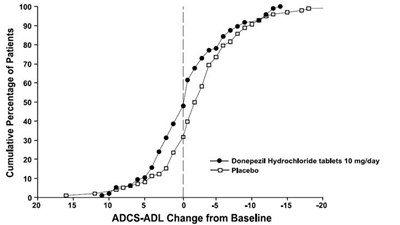
This is a chart that shows the cumulative percentage of patients who experienced a change in their ADAS-cog scores (a measure of cognitive function) after taking different doses of donepezil hydrochloride tablets (5 mg/day and 10 mg/day) or a placebo. The chart also shows the percentage of patients who experienced no change in their scores. The x-axis shows the change in ADAS-cog scores from baseline and the y-axis shows the cumulative percentage of patients.*
figure-04 - 30406488 figure 04
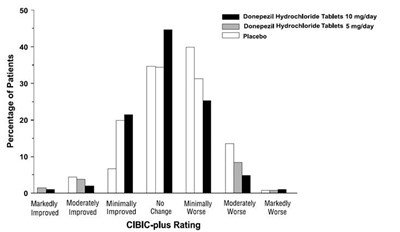
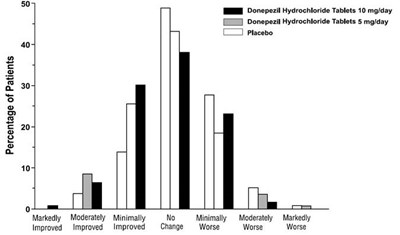
This is a chart showing the percentage of patients who took either Donepezil Hydrochloride Tablets 10mg/day, Donepezil Hydrochloride Tablets 5mg/day, or placebo. It also shows the CIBIC-plus Rating of the patients, with options ranging from markedly improved to markedly worse. No further context or information is given.*
figure-05 - 30406488 figure 05
The text is a chart or a table showing the ADAS-cog change from baseline that compares the clinical improvement among subjects who were given Donepezil Hydrochloride tablets in either 10mg/day or 5mg/day doses versus those who received a placebo. The change was measured over the course of 12 weeks of drug treatment. There is no quantitative data provided.*
figure-06 - 30406488 figure 06

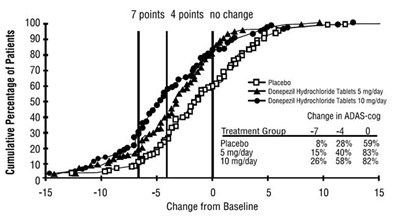
The given text seems to represent a table of data related to a medical study involving a treatment group and placebo group. The heading "Change in ADAS-cog" suggests it may be related to a measurement of cognitive function. However, the table is not completely readable or understandable, so specific details about the study and its results are not available.*
08 - 30406488 figure 08
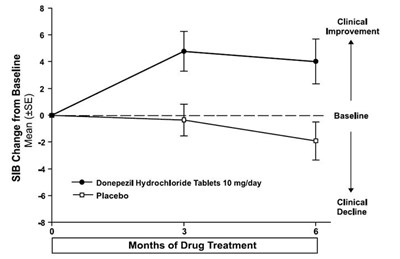
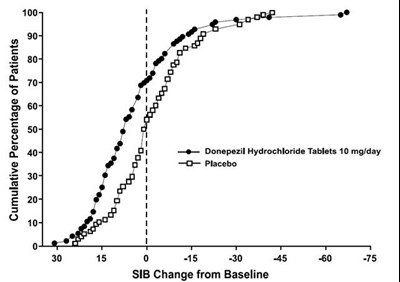
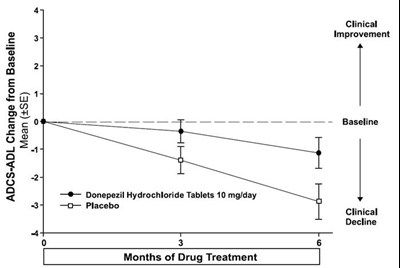
This is a comparison of the mean change from baseline in SIB scores for patients taking Donepezil Hydrochloride Tablets 10 mg/day versus placebo. The chart also indicates the level of clinical improvement and the duration of drug treatment (3 months).*
13 - 30406488 figure 13
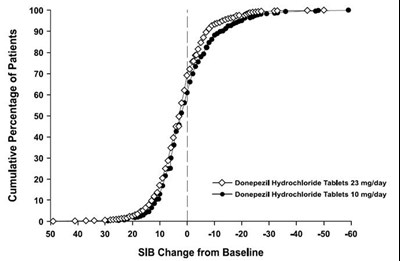
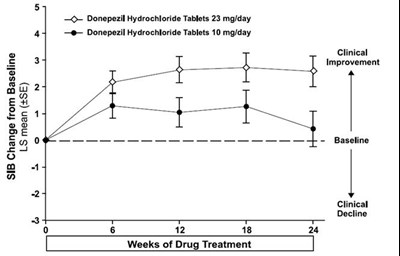
This is a graph that shows the cumulative percentage of patients taking two different doses of Donepezil Hydrochloride Tablets (10 mg/day and 23 mg/day) and their corresponding SIB (Severe Impairment Battery) Change from Baseline. The graph shows that a higher percentage of patients taking the 23 mg/day dose experienced an improvement in SIB scores compared to those taking the 10 mg/day dose.*
14 - 30406488 figure 14
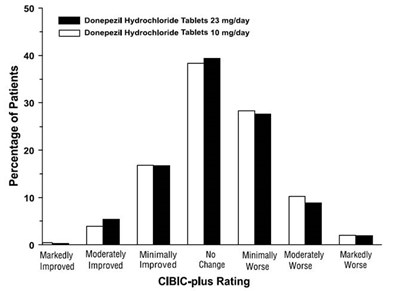
This appears to be a chart or graph with data about patients and their responses to Donepezil Hydrochloride Tablets at different dosages. The chart includes percentages of patients who showed varying degrees of improvement or decline in their condition, as well as a CIBIC-plus rating. Without more context, it is difficult to determine the exact purpose or significance of this data.*
16 - 30406488 figure 16

Donepezil is a medication available in the form of Hydrochloride Tablets, USP. The package contains 30 tablets, each of which comprises TOOF o 2ams.000.08 o e 23 mg. The drug is primarily used to treat certain mental/mood disorders like dementia. The medication should be kept in a light-resistant container at a moderate temperature, and it should not be dispensed if the package is broken or missing. The text also refers to some test results like the USP Dissolution Test and Organic Impurities Test. The contact address for further prescription information is Paramus, NJ 07652. The last line of text is a location reference in Taoyuan City, Taiwan.*
17 - 30406488 figure 17

Donepezil is a medication available in a bottle containing 90 tablets of hydrochloride. Each tablet contains 23mg of an active ingredient. The medication information and prescription details can be found on the packaging. It is recommended to store the tablets in a tamper-resistant, light-resistant container at room temperature. To ensure the effectiveness of the medication, dissolve the tablet and conduct a USP dissolution test. The company address is located in Paramus, NJ 07652 with another mention of an address in Taoyuan City 32063, Taiwan.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.