Product Images Pemetrexed
View Photos of Packaging, Labels & Appearance
- Structural formula - pem04 0000 01
- Figure 1 - pem04 0000 02
- Figure 2 - pem04 0000 03
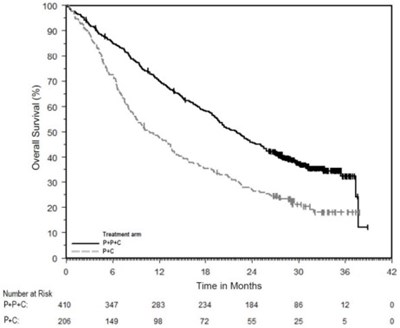
- Figure 3 - pem04 0000 04
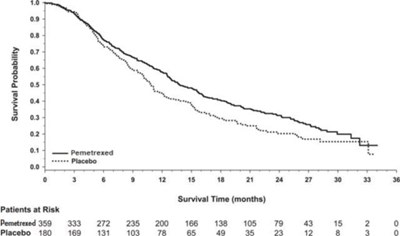
- Figure 4 - pem04 0000 05
- Figure 5 - pem04 0000 06
- Figure 6 - pem04 0000 07
- Figure 7 - pem04 0000 08
- Figure 8 - pem04 0000 09
- Figure 9 - pem04 0000 10
- PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label - pem04 0000 11
- PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label - pem04 0000 12
Product Label Images
The following 12 images provide visual information about the product associated with Pemetrexed NDC 25021-260 by Sagent Pharmaceuticals, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
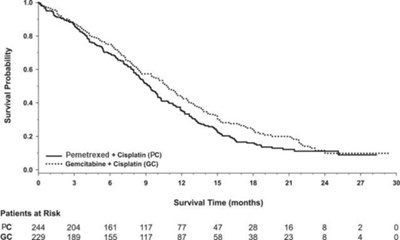
Figure 3 - pem04 0000 04

This text provides data on survival probability and survival time in months for two groups of patients: PC and GC. The number of patients at risk at different time points is also shown.*
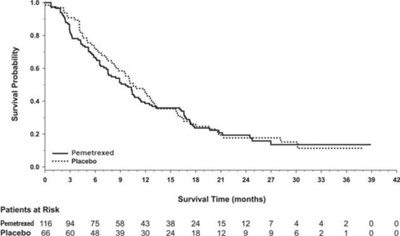
Figure 5 - pem04 0000 06

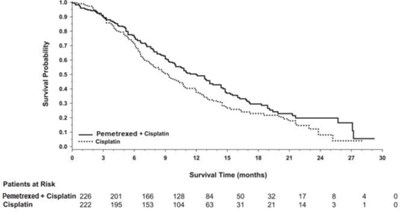
This text is about the survival probability of patients over time, comparing the treatment with Pemetrexed to a placebo. It includes the number of patients at risk at different time points in months. The data suggests that the treatment with Pemetrexed had more patients surviving at the observed time periods compared to those on the placebo.*
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label - pem04 0000 11

This text describes the contents of a single-dose vial intended for intravenous use only. It includes caution about it being a cytotoxic agent. The text also contains various codes, numbers, and references.*
PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Vial Label - pem04 0000 12

This is a label information for a product containing disodium equivalent to 500 mg. The product is manufactured for SAGENT Pharmaceuticals in Schaumburg, IL, USA and made in China. The label includes a copyright notice for Sagent Pharmaceuticals and a unique identification number.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.