Product Images Sildenafil Citrate
View Photos of Packaging, Labels & Appearance
- Sildenafil_fig1.jpg - Sildenafil fig1
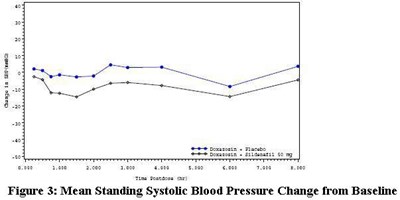
- Sildenafil_fig3 - Sildenafil fig 3
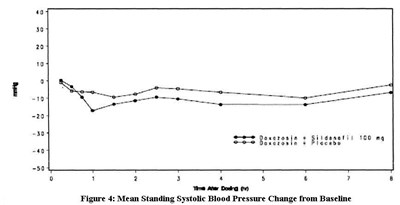
- Sildenafil_fig_4 - Sildenafil fig 4
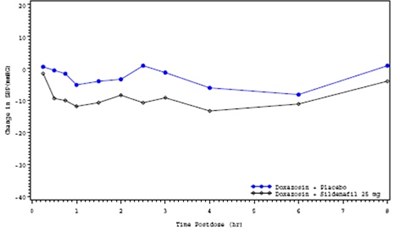
- sidenafil_fig_5 - Sildenafil fig 5
- sildenafil 100mg
- sildenafil 25mg
- sildenafil 50mg
- sildenafil-fig7 - sildenafil fig7
- str - sildenafil structure
- figure2 - sildenafil Fig2
- sildenafil_fig6b - sildenafil fig6b
- sildenafil_fig6a - sildenafil figure6a
Product Label Images
The following 12 images provide visual information about the product associated with Sildenafil Citrate NDC 27241-069 by Ajanta Pharma Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
sidenafil_fig_5 - Sildenafil fig 5
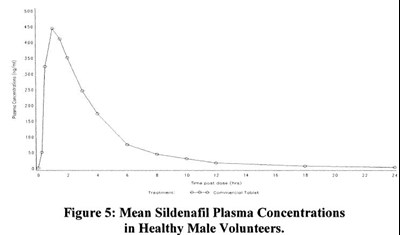
This is a figure showing the average levels of sildenafil in the blood of healthy men who have taken the drug.*
sildenafil 100mg
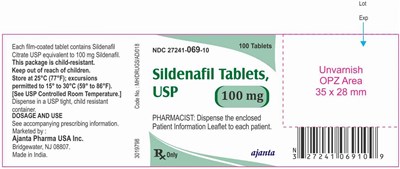
This is a package of Sildenafil Citrate tablets containing 100 mg of Sildenafil in each film-coated tablet. The package is child-resistant and should be kept out of the reach of children. It should be stored at room temperature between 15°C to 30°C. Dispense in a child-resistant container. The dosage and use information can be found in the accompanying prescribing information. This product is marketed by Ajanta Pharma USA Inc. in Bridgewater, NJ, and made in India. The package contains 100 tablets and the NDC number is 27241-069-10. The pharmacist should give the enclosed Patient Information Leaflet to each patient.*
sildenafil 25mg
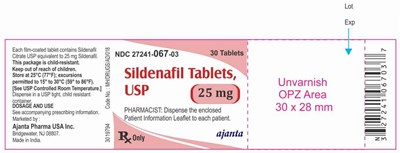
This is a description of a medication called Exp. It comes in the form of film-coated tablets containing Sildenafil and Citrate USP equivalent to 25 mg Sildenafil. The product should be stored between 15° to 30°C and kept in a child-resistant package. The recommended dosage and use information is not available. The medication is marketed by Ajanta Pharma USA Inc., and made in India. The package contains 30 tablets and has an NDC number of 27241-067-03. There is a note for pharmacists to disperse the enclosed patient information leaflet to each patient.*
sildenafil 50mg
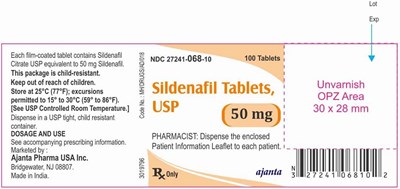
This is a package of 100 tablets of Sildenafil Citrate USP equivalent to 50 mg Sildenafil. It is child-resistant and should be stored at 25°C (77°F) with excursions permitted to 15° to 30°C (59° to 86°F). Dispensing should be done using a USP tight, child-resistant container. Dosage and use information can be found in the accompanying prescribing information leaflet, which should be given to each patient. This product was made in India and is marketed by Ajanta Pharma USA Inc. The NDC code is 27241-068-10.*
sildenafil-fig7 - sildenafil fig7
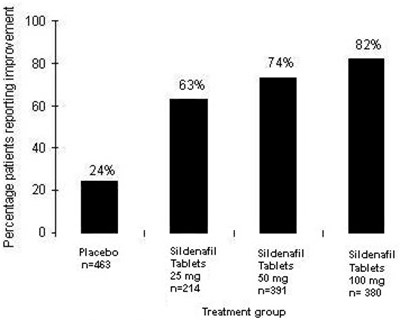
This is a chart showing the percentage of patients reporting improvement after taking different tablets. The chart includes a placebo and two other tablets labeled "Sidenail" and "Sideral" with dosages of 25mg. There is also another tablet labeled "Sitdari" with a 2% improvement rate.*
sildenafil_fig6a - sildenafil figure6a
Not available. The provided text is not readable as it seems to be a random string of characters.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.