Product Images Tadalafil
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Tadalafil NDC 27241-114 by Ajanta Pharma Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
tadalafil 10mg

Each tablet of Tadalafil USP 10 mg contains FD&C Yellow No. 5 (Tartrazine) as a color additive. The usual dosage should be checked in the accompanying literature. It is warned to keep the tablets out of reach of children and not to split them. The tablets should be taken in their entirety. The medication should be dispensed in a tight container and stored between 20° to 25°C (68° to 77°F) with permitted excursions of 157 to 30°C (59° to 86°F) as per USP Controlled Room Temperature. These tablets are made in India and marketed by Ajanta Pharma USA Inc. 30 tablets are included in each pack having NDC 27241-113-03.*
tadalafil 2 5mg

This is a medication with the active ingredient Tadalafil, USP 2.5 mg, available in film-coated tablets. The usual dosage is one tablet daily, and it should be stored in a tight container at 20° to 25°C (68° to 77°F), with excursions permitted to 15° to 30°C (59° to 86°F). The tablets contain FD&C Yellow No. 5 (Tartrazine) as a color additive. The NDC is 27241-111-03 and it is marketed by Ajanta Pharma USA Inc. The code number is GUJ/DRUGSIGI25/2080. The medication is not recommended for children, and the entire dose should be taken. The accompanying literature should be consulted for dosage information.*
tadalafil 20mg

This is a product description for Tadalafil tablets, which are coated and contain 20 mg of Tadalafil, a medicine used to treat erectile dysfunction (ED) or pulmonary arterial hypertension (PAH). The tablets should be taken as a whole and not be split. The usual dosage recommendations can be found in the accompanying literature. The product contains FD&C yellow No.5 (Tartrazine), and it should be dispensed in a tight container and stored at a temperature of 20° to 25°C (68° to 77°F), with excursions permitted to 15° to 30°C (59° to 86°F). The product is manufactured by Ajanta Pharma in India and distributed by Ajanta Pharma USA Inc. in Bridgewater, NJ.*
tadalafil 5mg

Each tablet of Tadalafil USP 5mg contains Tartrazine as a color additive. The usual dosage is mentioned in the accompanying literature. The tablets should not be split and the entire dose must be taken. The tablets are dispensed in a tight container and should be stored between 20°C to 25°C (68°F to 77°F) with excursions allowed between 15°C to 30°C (59°F to 86°F). The product should be kept out of the reach of children. The tablets are marketed by Ajanta Pharma USA Inc. and made in India. The product has an NDC code of 27241-112-03 and a Gode No. of GUJIDRUGS/G/25/2080.*
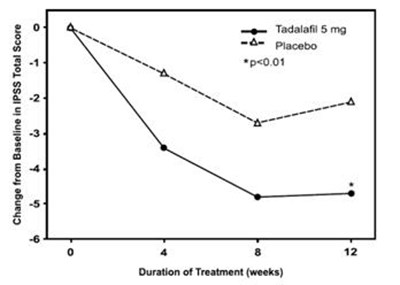
tadalafil fig8

This is a report on the IPSS Total Score, which includes data on the change from baseline, as well as treatment with Tadalafil 5mg compared to a placebo. The results show a significant difference between the two treatments, with Tadalafil 5mg showing a lower score compared to placebo, with a p-value of less than 0.01. The duration of treatment, however, is not available.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.