Product Images Droxidopa
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Droxidopa NDC 27241-199 by Ajanta Pharma Usa Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
100mg - droxidopa 100mg

This is a medication package for Droxidopa containing 90 capsules, each capsule containing 100 mg of Droxidopa. It is important to refer to the package insert for prescribing information, however the usual dosage is not provided. The package is child-resistant and should be stored at 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). The capsules should be dispensed in a tight, light-resistant container and not used if the seal under the cap is broken or missing. A warning is provided to keep this medication out of reach of children. The product is marketed by Ajanta Pharma USA Inc. and made in India. The Lot, Exp date and code number are also provided.*
200mg - droxidopa 200mg

Each capsule of the medicine contains 200 mg of Droxidopa. It is recommended to refer to the package insert for recommended dosage instructions. The product should be stored in a child-resistant, tightly sealed, and light-resistant container at a temperature of 20°C to 25°C. The medication is available in a package size of 90 capsules. It is manufactured in India and marketed by Ajanta Pharma USA Inc., located in Bridgewater, NJ. The code number mentioned on the package is MHIDRUGS/AD/018. If the seal under the cap is broken or missing, do not use the medicine. A warning is given to keep it out of reach of children.*
300mg - droxidopa 300mg

Each capsule of Droxidopa contains 300 mg of the medicine. It is important to read the package insert for prescribing information regarding usual dosage. This package contains 90 capsules and is child-resistant. It should be stored at a temperature between 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). Droxidopa should be dispensed in a tight, light-resistant container and discarded if the seal is broken or missing. It is a medication that should be kept far from children's reach. This product has been manufactured in India and marketed by Ajanta Pharma USA Inc. This is the only information available from the text.*
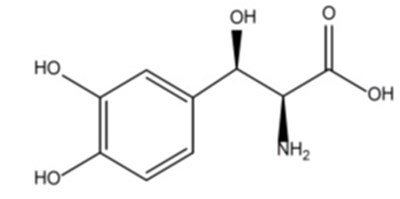
figure1 - droxidopa figure 1

This is a report of a clinical trial that evaluated the effectiveness of a drug called Droxidopa in reducing dizziness. The baseline value of dizziness was 5.1 in all participants, and the study had a double-blind treatment phase of 6 weeks. The mean change in dizziness after treatment with Droxidopa was not reported.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.