Product Images Doxazosin Mesylate
View Photos of Packaging, Labels & Appearance
- Image - 23feea8d ed70 456c 8426 e50332cea9c0 159 1
- Image - 23feea8d ed70 456c 8426 e50332cea9c0 174 2
- Chemical Structure - e2406e8e 68c2 49dc 9e15 6170f3fffa03 01
- Image - e2406e8e 68c2 49dc 9e15 6170f3fffa03 02
- Image - e2406e8e 68c2 49dc 9e15 6170f3fffa03 03
- Doxazosin Tablets USP, 1 mg - e2406e8e 68c2 49dc 9e15 6170f3fffa03 04
- Doxazosin Tablets USP, 2 mg - e2406e8e 68c2 49dc 9e15 6170f3fffa03 05
- Doxazosin Tablets USP, 4 mg - e2406e8e 68c2 49dc 9e15 6170f3fffa03 06
- Doxazosin Tablets USP, 8 mg - e2406e8e 68c2 49dc 9e15 6170f3fffa03 07
Product Label Images
The following 9 images provide visual information about the product associated with Doxazosin Mesylate NDC 29300-354 by Unichem Pharmaceuticals (usa), Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image - e2406e8e 68c2 49dc 9e15 6170f3fffa03 03

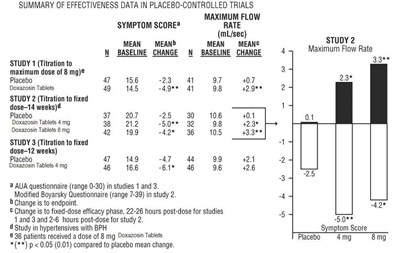
The text is a description of a study evaluating the mean change in total symptom score and increase in maximum urinary flow rate (in mL/sec) from baseline in a treatment group compared to a placebo. The study appears to have lasted for a certain number of weeks (not specified). The text also includes some statistical notation indicating significant differences between groups.*
Doxazosin Tablets USP, 4 mg - e2406e8e 68c2 49dc 9e15 6170f3fffa03 06

This is a description of a medication called Doxazosin Tablets. Each tablet contains 0.4 mg of doxazosin mesylate, a USP-approved ingredient. The recommended dosage can be found in the accompanying package insert. The tablets should be stored at a temperature of 20-25°C (68-77°F), with excursions permitted between 15-30°C (59-86°F). It is important to keep this medication and all drugs out of reach of children. The packaging contains 100 tablets and has a National Drug Code (NDC) of 29300-353-01. The manufacturer of this medication is Unichem Laboratories Ltd., located in Pilerne Ind. Estate, Bardez, Goa 403 511, India. The medication is manufactured for Unichem Pharmaceuticals (USA), Inc., located in East Brunswick, NJ 08816.*
Doxazosin Tablets USP, 8 mg - e2406e8e 68c2 49dc 9e15 6170f3fffa03 07

This text appears to be the description and details of a medication called Doxazosin. Each tabiet (tablet) contains 8 mg of doxazosin mesylate. The usual dosage is not mentioned and it is recommended to refer to the accompanying package insert for further instructions. The medication should be stored at a temperature range of 20°C to 25°C (68°F to 77°F) with limited excursions permitted between 15°C to 30°C (59°F to 85°F). It is important to keep this and all drugs out of reach of children. The text also includes the NDC number and manufacturer information, such as UNICHEM PHARMACEUTICALS and UNICHEM LABORATORIES LTD. The manufacturing location is mentioned as Pilerne Ind. Estate, Pileme, Bardez, Goa 403 511, India. Additionally, it is mentioned that the medication is manufactured for UNICHEM PHARMACEUTICALS (USA), INC. located in East Brunswick, NJ.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.