Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
- figr1
- figr2
- figr3
- logo1 - logo1
- logo2 - logo2
- Venlafaxine Hydrochloride 150mg 30 capsules - venlafaxine hydrochloride 150mg 30 capsules
- Venlafaxine Hydrochloride 30 capsules - venlafaxine hydrochloride 30 capsules
- Venlafaxine Hydrochloride 75mg 30 capsules - venlafaxine hydrochloride 75mg 30 capsules
- venlafaxinestructure - venlafaxinestructure
Product Label Images
The following 9 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 31722-003 by Camber Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figr1
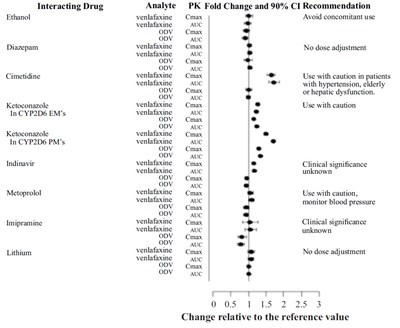
This text appears to be a list of drugs and their potential interactions with one another. The interacting drugs listed include ethanol, diazepam, cimetidine, ketoconazole, indinavir, metoprolol, imipramine, and lithium. The effects of these interactions on analytes, such as venlafaxine, are also listed, along with PK Fold Change and 90% CI Recommendation for Cmax and AUC. The recommendations for use vary from avoiding concomitant use to no dose adjustment, depending on the drugs involved and their interactions. Overall, this appears to be a reference guide for healthcare professionals when prescribing medication.*
figr2
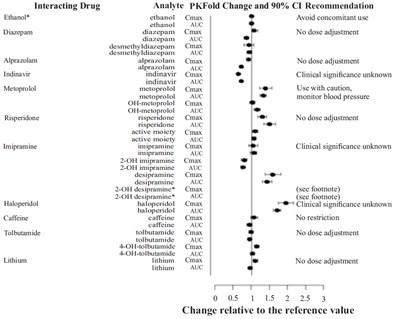
This appears to be a table of drugs and their interactions, including Cmax and AUC measurements for various analytes. The table provides recommendations for dose adjustment, caution, or avoidance of concomitant use for certain drug interactions. However, without further context, it is difficult to determine the specific purpose or application of this information.*
figr3
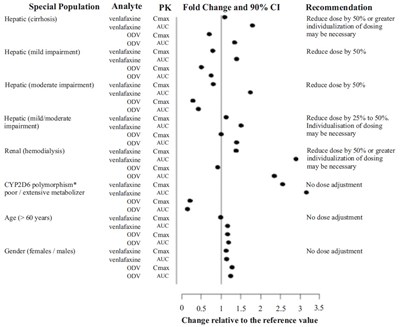
The text is a table showing PK (pharmacokinetics) recommendations for the use of venlafaxine in patients with different conditions such as hepatic or renal impairment, CYP2D6 polymorphism, age or gender. The table suggests dose adjustments or individualization of dosing based on the analyte and specific conditions. The last line indicates the change relative to the reference value.*
Venlafaxine Hydrochloride 30 capsules - venlafaxine hydrochloride 30 capsules

Each capsule contains Venlafaxine Hydrochloride for oral administration. The usual dosage of Venlafaxine Hydrochloride is 37.5mg. The medication comes in Extended-Release Capsules with 30 capsules per bottle. The product is manufactured for Cambir Pharmaceutical and should be dispensed with the prescribing information provided to each patient by the pharmacist. The prescription comes with Rxonly with an identification number of 2100434.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




