Product Images Amlodipine Besylate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Amlodipine Besylate NDC 31722-238 by Camber Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
8352ab43-figure-03 - 8352ab43 figure 03

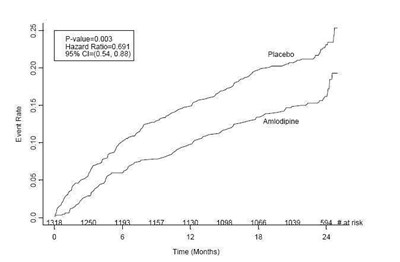
This text provides some information related to a study involving Amlodipine and Placebo. The results of the study favor Amlodipine over Placebo. The text also presents data related to the age and other characteristics of the participants in the study. Additionally, it mentions that the baseline SBP mean is 120 mmHg.*
8352ab43-figure-04 - 8352ab43 figure 04

This is a description of a medicine called Amlodipine Besylate Tablets, USP that comes in a pack of 90 tablets. Each tablet contains 2.5mg of Amlodipine Besylate. The medicine is only available by prescription and should be stored at a temperature between 20°C and 25°C. It should be protected from light and stored in a controlled room temperature. Details of the dosage and usage can be found in the accompanying prescribing information. The medicine is manufactured by Camber Pharmaceuticals in Fiscataway, NJ 06654, on behalf of InvaGen Pharmaceuticals, Inc. based in Hauppauge, NY 11785.*
8352ab43-figure-05 - 8352ab43 figure 05

This is a description of Amlodipine Besylate Tablets, USP by Camber Pharmaceuticals. The medication comes in a container of 90 Tablets which should be stored at controlled room temperature not exceeding 25°C (66°-77°F) and needs to be protected from light. It is advised to dispense in light-resistant containers. The given information does not include details on the dosage and use, but the accompanying prescription should have the details. Each tablet contains amlodipine besylate, which is equivalent to 5 mg amlodipine. Camber Pharmaceuticals, Inc. manufactures the medication, and it is manufactured by InvaGen Pharmaceuticals, Inc. located in Hauppauge, NY 11788.*
table1 - tab1

This appears to be a list of symptoms and their corresponding occurrence rates during clinical trials of a drug called Amlodipine administered in varying doses, as well as a placebo group. The symptoms listed are edema, dizziness, flushing, and palpitation. The numbers below indicate the number of participants in each group (N), percentage of occurrence of the symptom at each dose, and the corresponding percentage of occurrence in the placebo group.*
table2 - tab2

This is a comparison of the side effects experienced by people who took Amlodipine besylate tablets versus those who took a placebo. The data shows the percentage of individuals who reported experiencing specific side effects, including fatigue, nausea, abdominal pain, and somnolence. The sample size for those who took Amlodipine besylate tablets was 1730, while the sample size for the placebo group was 1250.*
table3 - tab3

This is a medical study that evaluated the effects of Amlodipine besylate tablets on both males and females. The data presented shows the percentage of males and females who experienced certain side effects such as edema, flushing, palpitations, and somnolence. The percentages of patients who experienced these symptoms are compared to a placebo group.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.