Product Images Lisdexamfetamine Dimesylate
View Photos of Packaging, Labels & Appearance
- 10 - 10
- 20 - 20
- 30 - 30
- 40 - 40
- 50 - 50
- 60 - 60
- Figure3 - Figure 3
- Figure1 - Figure1
- Figure2 - Figure2
- Figure4 - Figure4
- Figure5 - Figure5
- Figure6 - Figure6
- Figure7 - Figure7
- image description - Figure8
- chemical structure - chemical structure
- table1 - table1
- table2 - table2
- table3 - table3
- table4 - table4
- table5 - table5
- table6.1 - table6.1
- table6.2 - table6.2
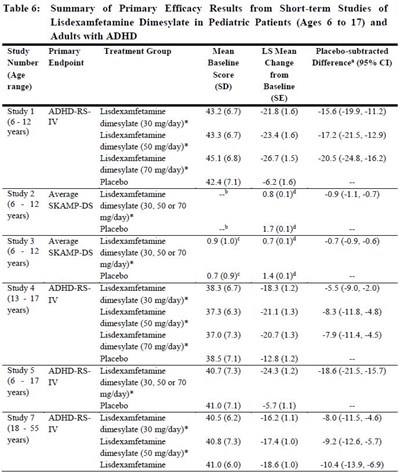
- table5.1 - table6
- table7 - table7
Product Label Images
The following 24 images provide visual information about the product associated with Lisdexamfetamine Dimesylate NDC 31722-321 by Camber Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 - 10

This text provides information about Lisdexamfetamine Dimesylate chewable tablets. Instructions include chewing the tablets completely before swallowing and storing at room temperature. It also mentions the importance of dispensing the Medication Guide to each patient. The tablets contain 10mg of Lisdexamfetamine Dimesylate equivalent to 5.3 mg of dextroamphetamine. Further dosage information can be found in the package insert.*
20 - 20

This is a description of Lisdexamfetamine Dimesylate Chewable Tablets. It provides instructions to chew the tablets completely before swallowing, advise for pharmacists to dispense the accompanying Medication Guide to each patient, dosage information, and storage instructions. The tablets contain Lisdexamfetamine dimesylate and are manufactured by Cambor Pharmaceuticals.*
30 - 30

This is a pharmaceutical product label for LisDEXAmfetamine Dimesylate chewable tablets, containing 30mg per tablet. It instructs to chew the tablets completely before swallowing and to not swallow the tablets whole. The text also advises pharmacists to dispense the accompanying Medication Guide to each patient. Storage instructions recommend keeping the tablets at room temperature. Additional information, such as prescribing details and a website for the Medication Guide, is provided.*
40 - 40

This is a label for a medication called Lisdexamfetamine Dimesylate Chewable Tablets. It contains instructions to chew the tablets completely before swallowing and advises against swallowing the tablets whole. Pharmacists are instructed to dispense a Medication Guide to each patient. The package contains 100 chewable tablets and should be stored at room temperature. Each tablet includes Lisdexamfetamine Dimesylate 40mg. More dosage information can be found in the package insert. A Medication Guide is available at timamberpharma.com/medicationguides.*
60 - 60

This text provides information about Lisdexamfetamine Dimesylate Chewable Tablets, including instructions for use, storage conditions, dosage information, and contact details of the manufacturer. It is important to chew the tablets completely before swallowing and not to swallow it whole. The medication guide should be dispensed to each patient with the product. The tablets contain 60 mg of Lisdexamfetamine Dimesylate. The manufacturer is Cambor Pharmaceutical located in Piscataway, NJ. Dosage information can be found in the package insert.*
Figure3 - Figure 3

This is a summary of the effects of Lisdexamfetamine Dimesylate on other drugs showing changes in drug levels such as Cmax (maximum concentration) and AUC (area under the curve) for various CYP enzymes and their substrates including Guanfacine, Venlafaxine, caffeine, Dextromethorphan, Omeprazole, and Midazolam. The data presented in Figure 3 indicates the fold change and 90% confidence intervals for these drug interactions.*
Figure1 - Figure1

This text provides valuable information on the impact of specific populations on the pharmacokinetics (PK) of d-amphetamine. It includes details on gender differences, age groups (comparing different age ranges to 55-64 years), and variations in PK parameters for renal impairment ranging from mild to end-stage renal disease. The text also highlights the PK fold change and 90% confidence intervals for Cmax and AUC, aiding in understanding how these populations may influence drug exposure and response.*
Figure2 - Figure2

This text provides information on the effect of other drugs on Lisdexamfetamine Dimesylate, showing changes in Cmax and Auc levels for Guanacine, Venlafaxine, and Omeprazole. The data is presented in a figure format and includes information on the fold change and confidence interval.*
Figure4 - Figure4

This is a graph showing the LS Mean SKAMP Deportment Subscale Score by Treatment and Time-point for Pediatric Patients Ages 6 to 12 with ADHD after 1 week of double-blind treatment (Study 3). The data includes scores for Placebo and Lisdexamfetamine dimesylate over different time points post-dose in hours. A higher score on the SKAMP-Deportment scale indicates more severe symptoms.*
Figure5 - Figure5

This text provides information on the Kaplan-Meier estimated proportion of pediatric patients aged 6 to 17 with treatment failure in Study 6. It compares the outcomes between patients treated with lisdexamfetamine dimesylate and those who received a placebo over a certain period of time. The data shows the proportion of subjects experiencing relapse or treatment failure at different time points (in days) after randomization, allowing for an analysis of treatment effectiveness over time.*
Figure6 - Figure6

This is a description of Figure 6 from a study (Study 8) showing the LS Mean (Standard Error) PERMP Total Score by Treatment and Time-point for adults aged 18 to 55 with ADHD after 1 week of double-blind treatment. The chart displays the LS Mean (SE) PERMP Total Scores over different time points (measured in hours) post-dose.*
Figure7 - Figure7

This text provides statistical information from Figure 7, showing the Kaplan-Meier Estimated Proportion of Subjects with Relapse in adults with ADHD for Study 9. The data compares the proportion of subjects with relapse/treatment failure between the placebo group and the group treated with Lisdexamfetamine dimesylate over a period of time (measured in days after randomization).*
image description - Figure8

This is a graphical representation of the Kaplan-Meier estimated proportions of subjects with relapse in adults with Binge-Eating Disorder (BED) from Study 13. The figure shows the proportion of subjects with relapse over time for individuals treated with Lisdexamfetamine dimesylate compared to a placebo. The x-axis represents time in days after randomization, while the y-axis displays the proportion of subjects with relapse. The data points indicate the percentage of relapse at specific time intervals. This figure provides valuable insights into the relapse rates in this particular study population.*
table1 - table1

This is a table showing the adverse reactions reported by pediatric patients ages 6 to 12 years with ADHD who were taking Lisdexamfetamine Dimesylate in a 4-week clinical trial. The table lists the percentage of patients experiencing each adverse reaction, with only those reactions reported by 2% or more of patients included in the table. Adverse reactions such as decreased appetite, insomnia, abdominal pain, irritability, vomiting, and weight decrease are highlighted in the table.*
table2 - table2

This is a table showing adverse reactions reported by pediatric patients aged 13 to 17 years with ADHD taking Lisdexamfetamine Dimesylate in a 4-week clinical trial. The percentages of these reactions are compared to those of the placebo group. Adverse reactions include decreased appetite, insomnia, weight decrease, dry mouth, palpitations, anorexia, and tremors.*
table3 - table3

This is a list of adverse reactions reported by 2% or more of adult patients with ADHD who took Lisdexamfetamine Dimesylate in a 4-week clinical trial. Some of the reported reactions include decreased appetite, insomnia, dry mouth, nausea, anxiety, restlessness, increased blood pressure, and increased heart rate.*
table4 - table4

This text provides a table showing adverse reactions reported by 2% or more of adult patients with BED (binge-eating disorder) taking Lisdexamfetamine Dimesylate, compared to those taking a placebo in 12-week clinical trials (Study 11 and 12). Common adverse reactions included dry mouth, insomnia, decreased appetite, increased heart rate, feeling jittery, constipation, anxiety, among others. The table also specifies the percentages of patients experiencing these adverse reactions for both Lisdexamfetamine dimesylate and placebo groups.*
table5 - table5

This text describes clinically important interactions between amphetamines and MAO Inhibitors (MAOI). The use of MAOI antidepressants can slow down amphetamine metabolism, leading to an increased effect on the release of norepinephrine and other monoamines, which can cause symptoms like headaches and signs of hypertensive crisis. Severe neurological effects and malignant hyperpyrexia can also occur, potentially resulting in fatal outcomes. It is advised not to administer lisdexamfetamine dimesylate during or within 14 days of MAOI administration to avoid these interactions.*
table5.1 - table6

This text provides detailed information on the clinical impact and interventions related to the use of lisdexamfetamine dimesylate with various medications such as serotonergic drugs, CYP2D6 inhibitors, alkalinizing agents, acidifying agents, and tricyclic antidepressants. It emphasizes the importance of monitoring patients for signs of serotonin syndrome and adjusting doses accordingly to avoid potential risks or interactions.*
table7 - table7

This is a summary table showing the Primary Efficacy Results in BED (Binge Eating Disorder) for two different studies with treatment using Lisdexamfetamine dimesylate compared to a placebo. The table includes information on the Treatment Group, Primary Efficacy Measure (Binge Days per Week at Week 12), Number of participants, Mean Baseline, LS Mean (Placebo-subtracted), Score Change with differences, and Confidence Intervals. It also provides information on standard deviation, standard error, and least-squares mean. The data indicates that the Lisdexamfetamine dimesylate treatment group (50 or 70 mg/day) showed statistically significant superiority over the placebo in reducing binge eating episodes.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.