Product Images Lisdexamfetamine Dimesylate
View Photos of Packaging, Labels & Appearance
- 10 - 10
- 20 - 20
- 30 - 30
- 40 - 40
- 50 - 50
- 60 - 60
- 70 - 70
- Figure 1 - Figure 1
- Figure 2 - Figure 2
- Figure 3 - Figure 3
- Figure 5&6 - Figure 5
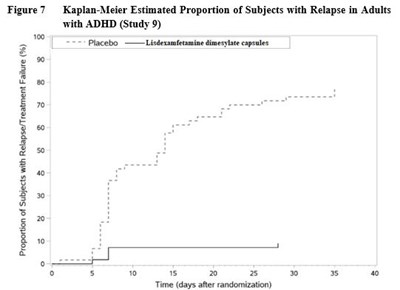
- Figure 7 - Figure 7
- Figure 8 - Figure 8
- MG3 - MG3
- MG4 - MG4
- MG5 - MG5
- MG6 - MG6
- MG7 - MG7
- MG8 - MG8
- MG9 - MG9
- structure - structural formula
- table12 - table 1 2
- table3 - table 3.1
- table4 - table 4
- table 5.1 - table 5.1
- table 5.2 - table 5.2
- table6.1 - table 6.2
- table6 - table 6
- table 7 - table 7
Product Label Images
The following 29 images provide visual information about the product associated with Lisdexamfetamine Dimesylate NDC 31722-355 by Camber Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 - 10

This text contains information about a medication named Lisdexamfetamine Dimesylate in 10 mg capsules. It instructs the pharmacist to dispense the enclosed Medication Guide to each patient. The storage conditions are also provided, with a recommendation to store at a temperature between 20°C-25°C (68°F-77°F). The text also mentions that each capsule contains Lisdexamfetamine Dimesylate equivalent to 5.3 mg of dexamfetamine. For more details, it advises to see the package insert for dosage information. The medication is manufactured by Ascont Pramacouica and for CamiberPraceutical.*
20 - 20

This is a description of a medication called Lisdexamfetamine Dimesylate in capsule form with a strength of 20 mg. The pharmacist is advised to dispense the enclosed Medication Guide to each patient. The medication is to be stored at room temperature between 20°C to 25°C (68°F to 77°F). The package insert should be referred to for dosage information. Each capsule contains Lisdexamfetamine Dimesylate equivalent to 11.6 mg of dexmethylphenidate. The medication is manufactured by Ascent Pharmaceuticals in Central Islip, NY, and distributed by Camber Pharmaceutical in Piscataway, NJ.*
30 - 30

This is a description of a medication called Lisdexamfetamine Dimesylate in capsule form with a dosage of 30 mg per capsule. The pharmacist is advised to provide a medication guide to each patient who is prescribed this medication. The packaging contains 100 capsules for pharmacy use only. It needs to be stored at room temperature between 20°C and 25°C (68°F to 77°F). The medication is manufactured by Aocont Pharmaceuticals and marketed by Camber Pharmaceuticals. For detailed dosage information and other instructions, it is recommended to refer to the package insert.*
40 - 40

This text is a description of a medication called Lisdexamfetamine Dimesylate in capsule form with a strength of 40 mg. It includes instructions for the pharmacist to disperse the enclosed Medication Guide to each patient. The medication should be stored at room temperature between 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F). The package insert should be consulted for dosage information. The medication is manufactured by Aurobindo Pharma and distributed by Camber Pharmaceuticals. For more information, the Medication Guide is available at camberpharma.com/medication-guides.*
50 - 50

This text provides information about temperature storage instructions for a product, possibly a medication. It mentions the temperature range for storage, permissible excursions beyond this range, and the type of controlled room required. Additionally, it includes details about the container, medication, dosage, and manufacturing information. Overall, it seems to be pharmaceutical-related documentation.*
Figure 1 - Figure 1

This is a description of special population characteristics such as gender, age groups (65-74 years and 75 years or older), renal impairment levels (mild, moderate, severe, end-stage renal disease), and PK parameters (fold change and 90% confidence interval). The text also includes a table displaying change relative to reference values at different points (10, 15, 20, 25).*
Figure 2 - Figure 2

This is a table showing changes in Cmax and AUC for Guanfacine, Venlafaxine, and Omeprazole relative to a reference measurement. The changes are expressed in terms of PK Fold Change and 90% confidence intervals. The table provides data points at different time intervals.*
Figure 3 - Figure 3

This description provides information on the interaction of various drugs with different cytochrome P450 enzyme substrates such as Guanfacine, Venlafaxine, Caffeine, Dextromethorphan, Omeprazole, and Midazolam. It also mentions a fold change and 90% confidence interval related to PK parameters, and a change relative to a reference value.*
Figure 5&6 - Figure 5

This text provides figures illustrating the treatment failure rates for pediatric patients aged 6 to 17 in Study 6 and the PERMP total score by treatment and time-point for adults aged 18 to 55 with ADHD after 1 week of double-blind treatment in Study 8. The PERMP scale is used to indicate the severity of symptoms, where a higher score suggests less severe symptoms.*
MG3 - MG3

This text provides important information for individuals who are pregnant or planning to become pregnant while taking Lisdexamfetamine dimesylate. It mentions the existence of a pregnancy registry to monitor the health of females and their babies exposed to the medication. If pregnancy occurs during treatment, individuals are encouraged to speak with their healthcare provider and consider registering with the National Pregnancy Registry for Psychostimulants. Contact information and a website link for the registry are provided for further guidance.*
MG4 - MG4
This text mentions various medications used for different purposes such as treating migraine headaches, depression, anxiety, and pain management. It includes selective serotonin reuptake inhibitors (SSRIs), serotonin norepinephrine reuptake inhibitors (SNRIs), triptans, tricyclic antidepressants, lithium, fentanyl, tramadol, tryptophan, buspirone, and St. John's Wort. These medicines are commonly used in medical practices for their therapeutic effects.*
MG5 - MG5
This description highlights circulation problems in fingers and toes, specifically peripheral vasculopathy such as Raynaud's phenomenon. Symptoms may include numbness, coolness, and pain in the fingers or toes, as well as changes in color from pale to blue to red. This information can help individuals recognize potential circulatory issues in their extremities.*
MG6 - MG6

This text appears to be a list of symptoms associated with a medical condition or adverse drug reactions. Symptoms include agitation, flushing, coma, loss of coordination, dizziness, hallucinations, high body temperature, fast heartbeat, seizures, sweating, confusion, tremors, muscle stiffness or twitching, changes in blood pressure, and gastrointestinal symptoms such as nausea, vomiting, and diarrhea. These symptoms should be promptly evaluated by a healthcare provider.*
MG7 - MG7

This text includes a list of common side effects or symptoms that may occur, such as loss of appetite (anorexia), decreased appetite, diarrhea, dry mouth, trouble sleeping, stomach pain, anxiety, weight loss, dizziness, irritability, nausea, and vomiting. These could be potential indications of a health condition, medication side effects, or other factors that may require attention.*
MG8 - MG8
This text seems to describe common side effects or symptoms associated with a specific condition or medication. Some of the mentioned symptoms include dry mouth, trouble sleeping, decreased appetite, increased heart rate, constipation, feeling jittery, and anxiety. These symptoms may be indicators of a health issue or adverse reactions to a treatment. It is advisable to consult a healthcare professional for further evaluation and guidance.*
MG9 - MG9

This text provides instructions on how to take Lisdexamfetamine dimesylate capsules. It explains that the capsules may be swallowed whole or the contents can be sprinkled onto yogurt, or mixed into water or orange juice if the capsule cannot be swallowed whole. The text advises breaking apart any stuck-together powder and stirring the mixture until well combined, then swallowing it right away. It also notes that it is normal to see a filmy coating on the inside of the glass or container after consuming the mixture.*
table12 - table 1 2

This text provides a summary of adverse reactions reported by pediatric patients with ADHD aged 6 to 12 years and 13 to 17 years while taking Lisdexamfetamine Dimesylate capsules compared to a placebo in separate 4-week clinical trials. Common adverse effects in the younger age group included decreased appetite, upper abdominal pain, and nausea. In contrast, the older age group also experienced decreased appetite, dry mouth, and palpitations. Overall, the results provide insights into the side effects associated with this medication in these age groups.*
table3 - table 3.1
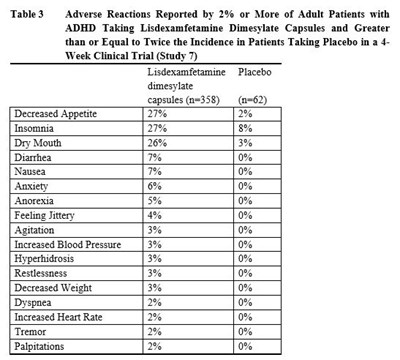
This is a table showing the adverse reactions reported by 2% or more of adult patients with ADHD taking Lisdexamfetamine Dimesylate Capsules, with a comparison to the incidence in patients taking a placebo in a 4-week trial. Common adverse reactions include decreased appetite, insomnia, dry mouth, diarrhea, and nausea. The percentage of patients experiencing each side effect is also provided for both the Lisdexamfetamine dimesylate capsules group and the placebo group.*
table4 - table 4

This data table provides a list of adverse reactions reported by adult patients taking Lisdexamfetamine Dimesylate capsules for Binge Eating Disorder (BED) in 12-week clinical trials. The table lists reactions that occurred in 2% or more of patients and at least twice as frequently compared to those taking a placebo. Common reactions include dry mouth, insomnia, decreased appetite, increased heart rate, feeling jittery, constipation, anxiety, and diarrhea. Less common reactions include vomiting, paresthesia, pruritus, and upper abdominal pain. Notably, there were no reported cases of hyperhidrosis, urinary tract infections, nightmares, oropharyngeal pain, increased energy, or restlessness.*
table 5.1 - table 5.1
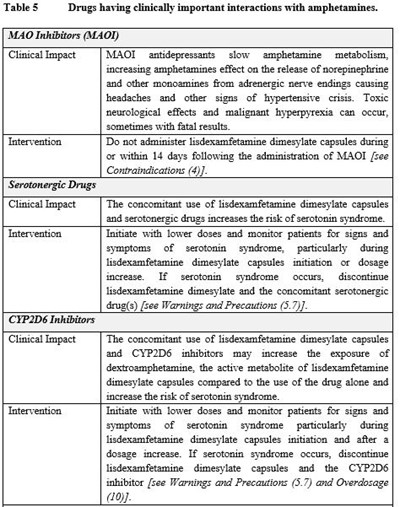
This text provides essential information on drugs that have clinically important interactions with amphetamines, specifically focusing on MAO inhibitors, serotonergic drugs, and CYP2D6 inhibitors. MAO inhibitors can slow amphetamine metabolism, potentially leading to hypertensive crises and fatal results if not used properly. On the other hand, serotonin syndrome risk increases when combining lisdexamfetamine dimesylate capsules with serotonergic drugs. Similarly, interactions with CYP2D6 inhibitors can increase dextroamphetamine exposure, raising the risk of serotonin syndrome. Lower doses and vigilant monitoring of patients for signs of serotonin syndrome are recommended to manage these interactions effectively.*
table 5.2 - table 5.2

This text is a list of clinical impacts and interventions related to drug interactions with urinary alkalinizing agents, acidifying agents, and tricyclic antidepressants. It highlights how these agents can affect the blood levels and efficacy of amphetamines, with recommendations to avoid certain combinations or adjust doses based on clinical response. Monitoring and adjusting treatment based on clinical response is emphasized for tricyclic antidepressants, as they can enhance the activity of certain agents and have potent cardiovascular effects.*
table6.1 - table 6.2

This text seems to be a summary or report of a study related to the treatment of Pediatric Patients Ages 6 to 12 with ADHD using Lisdexamfetamine dimesylate capsules. The study compares the effects of the treatment with Placebo and the medication in terms of SKAMP Deportment Scores. The SKAMP-Deportment scale measures symptom severity, and the text includes figures showing the results after 1 week of double-blind treatment. It seems to provide comparisons of scores between different treatment groups and time-points post-dose. The study appears to focus on the efficacy and safety of the medication in managing ADHD symptoms in pediatric patients.*
table 7 - table 7

This is a summary of primary efficacy results for binge eating disorder (BED) trials involving Lisdexamfetamine dimesylate. The table includes baseline data, standard deviations, standard errors, least-squares means, and confidence intervals for studies 11 and 12. The results show that Lisdexamfetamine dimesylate was statistically significantly superior to placebo in reducing binge eating symptoms.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.