Product Images Entecavir
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Entecavir NDC 31722-833 by Camber Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label1 - Label1

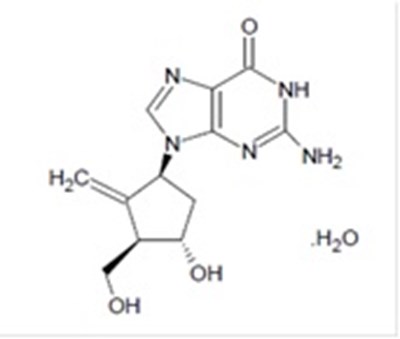
Entecavir is a medication available in tablet form with each film coated tablet containing 0.5mg of the medication. It is prescribed as per the package insert information. It should be stored in a tightly closed container at a temperature between 20°C to 25°C. The medicine is manufactured by Camber Pharmaceuticais and Hetero Labs Limited. The contact number for the US is 866-495-1995. No information is available for batch coding in this document.*
label4 - label4

This appears to be a pharmaceutical label for Entecavir tablets containing 0.5 mg of the medication. The tablets are manufactured by Camber Pharmaceuticals and Hetero Labs Limited. The NDC code is 31722-833.31. The rest of the text is not readable and contains errors from .*
label5 - label5

This is a description of a medication called Entecavir. It comes in the form of tablets and each tablet contains 1 mg of Entecavir. The usual dosage information can be found in the package insert. The medication should be stored in a tightly closed container at a temperature between 20-25°C. The manufacturer is Camber Pharmaceuticals, Inc. and the medication is distributed by Hetero Labs Limited, based in India. The text also includes some identification codes and contact information.*
label6 - label6

Entecavir is a medication provided in tablet form. Each tablet contains 1 g of encav. The recommended dosage should be obtained from the package insert. It should be stored in a tightly closed container at a temperature of 20 to 25 °C (68° to 77°F). The medication is manufactured for Camber Pharmaceuticals, and the US contact number is 866-495-1995. The medication is licensed under SUMNAPI2009/FR, and the batch coding should be present in an unvarnished area.*
label8 - label8

Description: The text appears to be a list of medication with the National Drug Codes (NDC) 31722-83431 assigned to the drug called "enlm:avil". The medication seems to come in the form of tablets produced by the pharmaceutical company "Cambor Pramacatica" under the trademark "Hetero" with an address listed as "Labs U, Unt". Other information in the text seems to be illegible and therefore not available for description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.