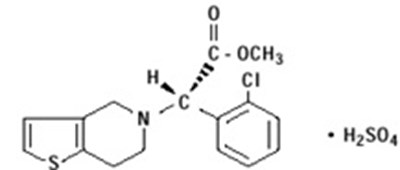
Product Images Clopidogrel
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Clopidogrel NDC 31722-902 by Camber Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Clopidogrel300mg30s - Clopidogrel300mg30s

This is a medication called Clopidogrel Tablet, which contains 391.5 mg of Clopidogrel bisulfate, USP equivalent to 300 mg of clopidogrel base. It is meant for adults, and the usual adult dosage is not provided in the text, but can be found in the package insert. The medication should be stored at a temperature between 20-25°C (68-77°F), and up to 30°C (86°F) is permitted. The manufacturer is Camber Pharmaceuticals, Inc. The text also includes some codes and contact information.*
Clopidogrel30s75mg - Clopidogrel30s75mg

The text describes the composition and dosage of Clopidogrel bisulfate tablets, which contains 97.875mg of the active ingredient, equivalent to 75mg of clopidogrel base. The recommended storage temperature range and contact information are provided. The drug is distributed in a package of 30 tablets and comes with a Medication Guide that should be provided to each patient. The manufacturer is Camber Pharmaceuticals, and the drug is produced by ScieGen Pharmaceuticals. The lot and expiration dates are also mentioned.*
Clopidogrelfigure2 - Clopidogrelfigure2

This document appears to be a medical report or record, specifically detailing cases of cardiovascular death, myocardial infarction (heart attack) and stroke. The location mentioned is "Wyocarta" and there is a reference to a "CURE Sty" which may indicate a treatment or intervention program. The document is labeled "PLACESO (1 cpi)".*
figure5 - figure5

This appears to be a description of a clinical trial comparing the efficacy of a placebo versus a medication called Clopidogrel in reducing the risk of death, re-infarction (the formation of a new blood clot) or stroke before a patient's first discharge. The results show that a smaller percentage of patients taking Clopidogrel experienced an event compared to those taking the placebo, with a 9% proportional risk reduction that was statistically significant (p=0.002). The timeline for the study extended up to 28 days since the start of the trial.*
figure8 - figure8

The text describes a chart or a table showing the cumulative event rate (% of fatal or non-fatal vascular events) of Clopidogrel Bisulfate after 22 months of follow-up. There are percentages listed for time periods of 0, 12, 18, and 22 months.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.