Product Images Aripiprazole
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 27 images provide visual information about the product associated with Aripiprazole NDC 31722-920 by Camber Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10mg - 10mg

Each tablet of this medication contains 10mg of aripiprazole. The usual dosage should be checked through the package insert. The medication should be stored in tightly closed containers at a controlled room temperature. The medication is manufactured by Camber Pharmaceuticals, Inc. in Piscataway, NJ. The dispensing information is provided separately to each patient by Ascent Pharmaceuticals, Inc. This medication is available in tablet form.*
15mg - 15mg

This is a description of Aripiprazole Tablets, USP. Each package comes with 100 tablets, with each tablet containing 15 mg of Aripiprazole. The medication guide should be provided to each patient. Dosage instructions are included in the package insert. The tablets should be stored in tightly closed containers at room temperature. The manufacturer is Camber Pharmaceuticals, Inc., located in Piscataway, NJ 08854, and the medication is manufactured by Ascent Pharmaceuticals, Inc. in Central Islip, NY 11722.*
20mg - 20mg

This is a description of an antipsychotic medication called Aripiprazole. The medication is available in tablet form with each tablet containing 20 mg of Aripiprazole USP. The usual dosage information is given in the package insert. The tablet should be stored in tightly closed containers at room temperature. The medication is manufactured by Ascent Pharmaceuticals and marketed by Camber Pharmaceuticals. The address and contact details of the manufacturers are provided.*
2mg - 2mg

This is a description of medication named "Aripiprazole Tablets, USP". Each tablet contains 2mg of the medicine. It is usually prescribed by a doctor and packaging leaflet may provide more details. The medication is manufactured by Ascent Pharmaceuticals, Inc. in Central slip, NY 11722, and distributed by Camber Pharmaceuticals, Inc. located in Piscataway, NJ 08854. It should be stored in tightly closed containers at a controlled room temperature.*
30mg - 30mg

This is a description of Ariiprazole Tablets, USP which contain 30mg of Ariiprazole USP. It contains information on usual dosage and how to store the tablets in tight closed containers at room temperature. It is manufactured by Ascent Pharmaceuticals, Inc. and distributed by Camber Pharmaceuticals, Inc. in Piscataway, NJ.*
5.4 - 5.4

This text describes the results of clinical trials testing the efficacy of fixed doses of aripiprazole tablets in treating schizophrenia. The trials were conducted over 4-6 weeks with varying sample sizes and doses of medication compared to a placebo. Results show that aripiprazole tablets were superior to placebo in improving PANSS and CG1-severity scores, with some doses also showing improvement in PANSS positive and negative subscales. The 10mg dose was the most effective on the primary outcome measure of the trials.*
5.7 - 5.7

The text discusses post-marketing case reports indicating that patients taking aripiprazole may experience intense urges, including gambling, sexual urges, shopping, eating or binge eating, and other impulsive or compulsive behaviors. Prescribers should ask patients or caregivers specifically about the development of such urges as patients may not recognize them as abnormal. Compulsive behaviors may result in harm, so it is important to consider dose reduction or stopping the medication if a patient develops such urges.*
5.9 - 5.9
The text provides cautions and instructions for healthcare professionals regarding the potential side effects of antipsychotic medication, specifically aripiprazole tablets. These side effects, including somnolence, postural hypotension, and motor and sensory instability, can increase the risk of falls and resulting injuries, especially in patients with certain medical conditions or who are taking certain medications. The text recommends that healthcare providers perform fall risk assessments when initiating antipsychotic treatment and periodically for patients on long-term antipsychotic therapy.*
5mg - 5mg

This is a medication bottle label for Aripiprazole tablets, with a dosage of 5mg per tablet and 100 tablets in the bottle. The usual dosage instructions are not provided on the label and should be obtained from the package insert. The medication should be stored at room temperature in tightly closed containers. The medication is manufactured by Ascent Pharmaceuticals and distributed by Camber Pharmaceuticals.*
Figure2 - Figure2

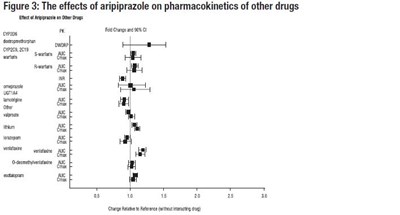
This is a figure, divided into two parts (Figures 1 and 2), presenting the effects of various drugs on the pharmacokinetics of aripiprazole and dehydro-aripiprazole. The first graph shows the fold change and 90% G for aripiprazole when interacting with several drugs, such as itraconazole, ketoconazole, quinidine, and carbamazepine. The second graph illustrates the effects of different drugs (ovandem, lamotrigine, omeprazole, etc.) on the pharmacokinetics of dehydro-aripiprazole. Both graphs use a relative change scale ranging from 0.5 to 3.0.*
Figure6 - Figure6
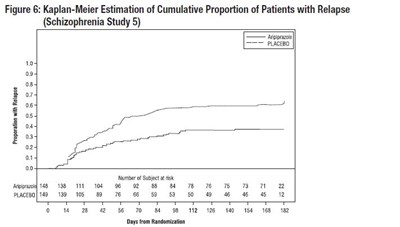
This is a Kaplan-Meier estimation graph that shows the cumulative proportion of patients with relapse in schizophrenia study 5. The graph tracks the proportion of patients with relapse over time (in days from randomization) and shows a comparison of results for the placebo and ipprzoe]LAGEHO treatments. The x-axis shows the number of days from randomization, while the y-axis shows the cumulative proportion of patients with relapse (ranging from 0% to 100%).*
mg3 - MG3

This is a safety warning advising individuals to monitor any changes in mood, behavior, thoughts or feelings when starting or adjusting the dosage of an antidepressant medication. It is important to report any sudden or new changes to the healthcare provider and attend all scheduled follow-up visits. In case of concerns about symptoms, it is recommended to contact the healthcare provider between visits.*
mg1 - mg1

This text is a warning label that accompanies a medication called aripiprazole. It cautions that the drug may cause increases in blood sugar which, in extreme cases, can lead to coma or death. Individuals with risk factors for diabetes should have their blood sugar checked before and during treatment. Symptoms of high blood sugar include excessive thirst, frequent urination, hunger, weakness, nausea, confusion, and fruity breath. The medication may also cause increased fat levels and weight gain, so patients are advised to monitor their weight regularly.*
mg2 - mg2

This is a list of the common side effects that can be experienced by adults and children who take aripiprazole tablets. These side effects include nausea, vomiting, constipation, headache, blurred vision, upper respiratory illness, dizziness, anxiety, insomnia, restlessness, inner sense of restlessness/need to move, feeling sleepy, fatigue, increased or decreased appetite, increased saliva or drooling, stuffy nose, weight gain, uncontrolled movement such as restlessness, tremor, and muscle stiffness.*
tab10 - tab10

The table provides information on the adverse reactions reported by adult patients treated with oral Aripiprazole tablets in short-term, placebo-controlled trials. The reactions reported by at least 2% of patients are mentioned. The table includes adverse reactions from various body systems such as the eye, gastrointestinal, musculoskeletal, connective tissue, nervous system, psychiatric, and respiratory system. Some of the commonly reported adverse reactions include blurred vision, nausea, constipation, fatigue, headache, dizziness, agitation, insomnia, and anxiety.*
tab11 - tab11

This table shows the percentage of patients experiencing adverse reactions in short-term, placebo-controlled trials of pediatric patients (6 to 18 years) treated with oral aripiprazole tablets. The table lists the preferred terms grouped by system organ class and reports the number of patients experiencing the term for both treatment groups: aripiprazole tablets and placebo. The adverse reactions reported by at least 2% of patients treated with aripiprazole tablets (excluding those with an incidence equal to or less than placebo) are mentioned. Some of the common adverse reactions include nausea, vomiting, diarrhea, weight increase, increased appetite, somnolence, headache, and sedation.*
tab12 - tab12

This is a medication guide that provides clinical rationale for using Aripiprazole tablets with other drugs. Aripiprazole has the potential to interact with strong CYP3A4 and CYP2D6 inhibitors, and strong CYP3A4 inducers, which can increase or decrease its exposure levels compared to when taken alone. This guide recommends monitoring the dosages while taking these drug combinations. Additionally, Aripiprazole has the potential to enhance the effect of certain antihypertensive agents, thus patients taking antihypertensive drugs are advised to monitor their blood pressure and adjust doses accordingly. The guide also explains that when Aripiprazole tablets are taken with benzodiazepines such as lorazepam, the intensity of sedation and orthostatic hypotension observed are greater compared to using Aripiprazole alone and advise adjusting doses accordingly.*
tab13 - tab13

This appears to be a table with information about different studies conducted on individuals with schizophrenia and the efficacy of different medications. The primary efficacy measure used was PANNS. The table shows the baseline scores, change from baseline, and differences for each treatment group. It also includes the standard deviation, standard error, least-squares mean, and confidence interval for each group. The table indicates that some doses of the medications were statistically superior to the placebo.*
tab6 - tab6
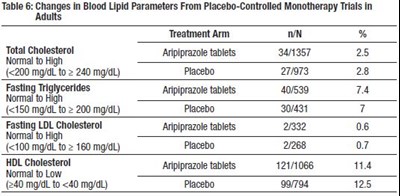
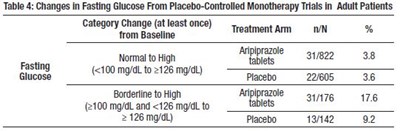
The table shows the changes in blood lipid parameters in adults treated with Aripiprazole tablets compared to Placebo in Monotherapy Trials. The table displays the percentage change in total cholesterol, fasting triglycerides, fasting LDL cholesterol, and HDL cholesterol with normal and high levels for each parameter. Unfortunately, there is not enough information to determine the units of measurement or the meaningfulness of the percentage changes.*
tab8 - tab8

The text is a table showing the percentage of patients experiencing weight gain of at least 7% of their body weight during placebo-controlled trials of adult patients. The table includes data for two different treatments (TN Apiprazole tablets and Avipiprazole tablets), as well as a placebo control group. The total number of patients in each treatment arm is also provided, along with the duration of the trials (either 6 weeks or 3 weeks).*
tab9 - tab9

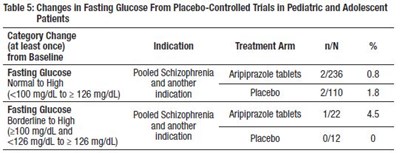
This is a table that shows the percentage of pediatric and adolescent patients who experienced weight gain greater than 7% of their body weight during placebo-controlled monotherapy trials. The trials involved the use of aripiprazole tablets in patients with schizophrenia and other indications, as well as a placebo. The table provides information on the number of patients in each treatment arm and the percentage of patients who experienced the specified weight gain. The trials lasted for six weeks.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.