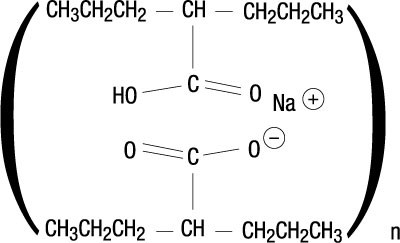
Divalproex Sodiumdelayed-release
Product Images NDC 33261-621
View Photos of Packaging, Regulatory Labels, and Product Appearance
Product Visual Gallery
This gallery contains 6 technical images submitted to the FDA as part of the official labeling for Divalproex Sodiumdelayed-release (NDC 33261-621). Unlike standard consumer photos, these assets often include clinical data figures, molecular chemical structures, and official manufacturer packaging layouts.
As provided by Aidarex Pharmaceuticals Llc, these visuals offer a comprehensive scientific overview of the product's physical and chemical identity, aiding pharmacists and researchers in product verification and study.
Product Images & Figures Index
Figure 1. Percentage Of Patients Achieving ≥ 30% Reduction In Symptom Score From Baseline (Divalproex Sod Dr 500mg Tab For Qualitest 2)

This figure shows the percentage of patients in Study 1 and Study 2 who achieved a greater than 30% reduction in symptom score from baseline. The results indicate that in both studies, 60% of patients treated with divalproex sodium (DVPX) were able to achieve this level of improvement. Placebo (PBO) was used as a comparison drug. The asterisk indicates that the results were statistically significant (p < 0.05).*
Figure 2 (Divalproex Sod Dr 500mg Tab For Qualitest 3)

The text represents a graph showing the reduction in CPS rate for patients taking Divalproex Sodium compared to placebo. The graph shows the percentage reduction in CPS rate ranging from -100% to 100%. The figure also includes a bar chart showing the percentage of patients who experienced improvement, no change, or worsening while taking Divalproex Sodium.*
Figure 3 (Divalproex Sod Dr 500mg Tab For Qualitest 4)

This is a graph (Figure 3) that shows the percentage reduction in CPS (Chronic Pain Score) rate for patients who were given either a high or low dose of a particular treatment. The graph shows that those who received the high dose experienced a greater reduction in CPS rate than those who received a low dose. The graph also shows the percentage of patients who experienced improvement, no change, or worsening of their CPS rate.*
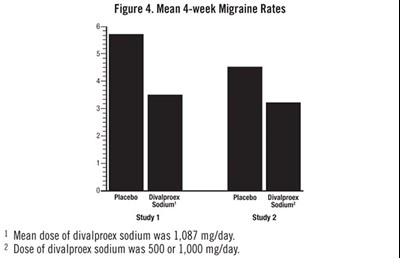
Figure 4. Mean 4-week Migraine Rates (Divalproex Sod Dr 500mg Tab For Qualitest 5)
Image Label (Divalproex Sod Dr 500mg Tab For Qualitest 6)

This package insert contains cautionary statements about dispensing the medication without a prescription and storing it in a controlled room. The medication contains Divalproex Sodium with 500mg strength and has 60 tablets in the bottle. The manufacturer and distributor of this medication are Aidarex Pharmaceuticals LLC and Qualitest Pharmaceuticals, respectively. The NDC number for this medication is 33261-0621-60. The package insert includes a chart with instructions for taking the medication.*
* These product label images have been analyzed using experimental machine learning. Please verify findings with the primary label text.