Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 26 images provide visual information about the product associated with Donepezil Hydrochloride NDC 33342-028 by Macleods Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
donepezil-10-mg-500 - donepezil 10 mg 500

Each tablet of MACLEODS contains 10 mg of Donepezil hydrochloride. It is a film-coated tablet that comes in a pack of 500 tablets with the batch number OPZ AREA. Its usage instructions can be found in the Prescribing Information section. The product must be stored at Controlled Room Temperature and comes with the Patient Information Leaflet. Macleods Pharma USA, Inc. manufactures the drug product, and it is produced by Macleods Pharmaceuticals Ltd., Baddi, Himachal Pradesh, India.*
donepezil-10 mg-30s - donepezil 10mg 30

This is a description of a drug product containing donepezil hydrochloride in the form of tablets USP. The manufacturer is Macleods Pharma USA, Inc. It is recommended to keep the product stored in controlled room temperature (20°-25°C or 68°-77°F). The usual dosage information can be found in the Prescribing Information Batch No. A Patient Information Leaflet should be dispensed along with the drug product. Batch No. is not available in the text.*
donepezil 23mg 10X10 - donepezil 23mg 10x10

Donepezil Hydrochloride Tablets is a medicine manufactured by Macleods Pharma USA in Plainsboro, New Jersey. The product comes in blister packs that aren't child-resistant with 100 tablets (10 x 10 unit-dose) in each pack. The dosage of the medication is specified in its prescribing information. It contains donepezil hydrochloride, USP 23 mg per film-coated tablet. The batch number and expiration date are G555 0s 01 and not specified, respectively. The product label states to store the pills in a controlled room temperature of 20-25°C (68-77°F), with permitted excursions to 15-30°C (50-86°F). A patient information leaflet accompanies the medicine.*
donepezil-23mg-30s - donepezil 23mg 30s

This is a description of a drug product called Donepezil Hydrochloride. It is a film-coated tablet that is prescribed in varying dosages and is used to treat certain mental conditions listed in its Prescribing Information. Users are advised to store the product at specific temperatures and with caution when transported. Manufactured by Macleods Pharma USA and Madeods Pharmaceutical Ltd of India, it needs to be dispensed with a Patient Information Leaflet by a pharmacist.*
donepezil 23mg 90s - donepezil 23mg 90s

This is a drug product called Donepezil Hydrochloride manufactured by Macleods Pharma USA. It comes in film-coated tablets of 23mg and is used for treating dementia. The package contains 90 Tablets, and it is advised to be stored at 20° to 25°C (68° to 77°F), with excursions permitted to 15° to 30°C (59° to 86°F). The product should be dispensed with the Patient Information Leaflet by the pharmacist. It is manufactured by Macleods Pharmaceuticals Ltd. in India.*
donepezil-5 mg-30s - donepezil 5 mg 30

This is a description of Donepezil hydrochloride tablets, produced by Macleod's Phorma USA, Inc. Each tablet contains 5mg of Donepezil hydrochloride USP, and it is a prescription drug with a recommended dosage as specified in the prescribing information. The tablets should be stored at a controlled room temperature of 20°-25°C (68°-77°F). The manufacturer is Macleods Pharmaceuticals Ltd, located in Baddi, Himachal Pradesh, India. The National Drug Code (NDC) is 33342-027-07, and the product may be dispensed with a patient information leaflet.*
donepezil-5-mg-500 - donepezil 5 mg 500

This is a description of a medication called Donepezil (brand name MR ponepezi) that comes in film-coated tablet form with a dosage of 5 mg. The tablets contain Phamalsa hydrochloride, USP. The recommended storage temperature is 20°-25°C (68>77°F). Additional information may be obtained from the prescribing information and patient information leaflet. The product is manufactured by Macleods Pharmaceuticals Ltd in India and distributed in the US by Macleods Pharma USA, Inc. The available package contains 500 tablets with code No.: HP/152/07 PMOT661601.*
donepezil ODT 10mg 30s - donepezil ODT 10mg 30s

This is a description of a medication called Donepezil Hydrochloride, which is available as orally disintegrating tablets USP of 10mg. The dosage and administration details are available in the prescribing information. The tablets come in a pack of 30 and should be stored at room temperature between 20°-25°C (68°-77°F). The medication is manufactured by Macleods Pharma USA, Inc, and the batch and expiry date details are available on the packaging. The product should be dispensed with a patient information leaflet by a pharmacist. The code number and manufacturing details of the product are also provided.*
donepezil ODT 10mg 4X7 - donepezil ODT 10mg 4X7
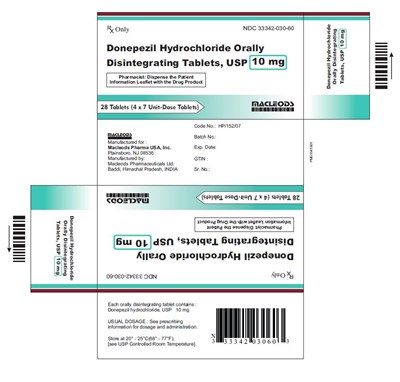
This is a package of donepezil hydrochloride orally disintegrating tablets, USP in a market presentation of 28 tablets (4 x 7 unit-dose tablets) with a concentration of 10 mg. The manufacturer is Macleods Pharma USA, Inc. in Plainsboro, NJ and the batch number is specified. It contains a patient information leaflet and should be stored at room temperature.*
donepezil ODT 5mg 30s - donepezil ODT 5mg 30s

This is a description of a medication called "Donepezil Hydrochloride". It comes in orally disintegrating tablets with 5mg of the medicine per tablet. The usual dosage is not specified, but prescribing information and instructions for dosage and administration can be found within the patient information leaflet. The medication was manufactured by Macleods Pharma USA, Inc. and Macleods Pharmaceuticals Ltd. and has a batch number of HP/152/07. The medication should be stored at a controlled room temperature between 20°C-25°C (68°F-77°F).*
donepezil ODT 5mg 4X7 - donepezil ODT 5mg 4X7
This is a description of a medication called Donepezil Hydrochloride Orally Disintegrating Tablets, USP 5 mg. It is dispensed with a Patient Information leaflet and comes in a blister pack of 28 tablets. The medication is manufactured by Macleods Pharmaceuticals Ltd in India and distributed by Macloods Pharma USA, Inc. in Plainsboro, New Jersey. The product code is HP152107 and the batch number is not legible. The normal dosage is not specified in the document, but should be obtained from prescribing information. The tablets should be stored at a controlled room temperature of 20° - 25°C.*
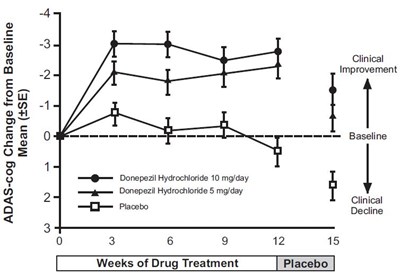
fig-1 - donepezil figure 1

The text is describing a graph which shows the ADAS-cog change from baseline with different drug treatments. The drugs mentioned are: Donepezil Hydrochloride 10mg/day, Donepezil Hydrochloride 5mg/day, and placebo. The x-axis shows the weeks of drug treatment while the y-axis shows the Mean (+ SE) improvement. The graph shows that both doses of Donepezil Hydrochloride resulted in some improvement in the ADAS-cog scores at different intervals compared to placebo.*
fig-10 - donepezil figure 10
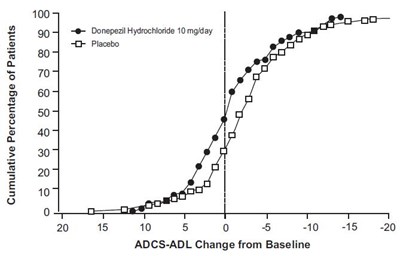
The text displays a graph showing the Cumulative Percentage of Patients for Donepezil Hydrochloride 10mg/day and Placebo, as well as an ADCS-ADL change from Baseline which appears to have a negative correlation with the time that has passed. It is likely a part of a medical or scientific report analyzing the efficacy of Donepezil Hydrochloride 10mg/day as a treatment for a certain condition.*
fig-11 - donepezil figure 11
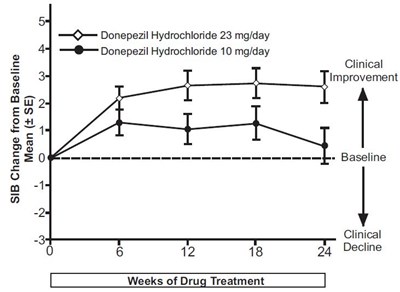
This is a graph that compares the effectiveness of a daily dose of Donepezil Hydrochloride at 23mg versus 10mg. The graph shows improvement in clinical symptoms of Alzheimer's disease over a period of 24 weeks. Not available.*
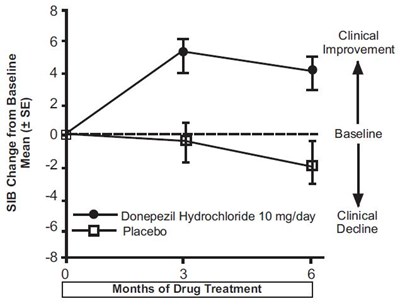
fig-12 - donepezil figure 12
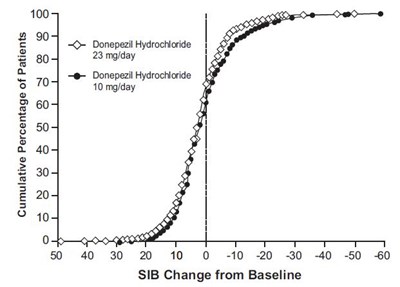
The text represents a chart showing the cumulative percentage of patients using Donepezil Hydrochloride at 23 mg/day and an unknown version. It also shows the change from baseline of the SIB (Severe Impairment Battery) score. Therefore, the chart possibly represents the effectiveness of the two versions of Donepezil Hydrochloride in treating patients with severe impairment.*
fig-13 - donepezil figure 13
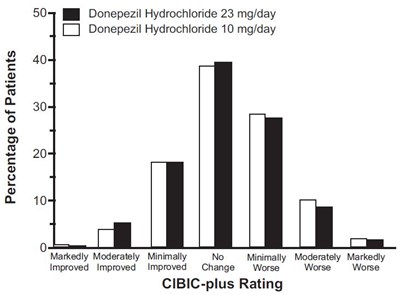
This appears to be a table or graph showing the results of a study on the effectiveness of two doses of Donepezil Hydrochloride (a medication commonly used to treat Alzheimer's disease) on the cognitive function of patients. The table shows the percentage of patients who experienced various levels of improvement or decline in their condition based on a CIBIC-plus rating scale, with one group receiving a dose of 23 mg/day and the other receiving a dose of 10 mg/day.*
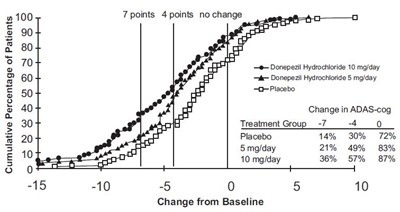
fig-2 - donepezil figure 2
This text appears to be a table or chart showing the effects of a drug called Donepezil on patients with ADAS-cog (Alzheimer's Disease Assessment Scale-cognitive subscale). The table shows the cumulative percentage of patients who experienced no change, a 4-point improvement, or a 7-point improvement in their ADAS-cog score after taking doses of 5mg or 10mg of Donepezil or a placebo. The table also indicates a change from baseline.*
fig-3 - donepezil figure 3
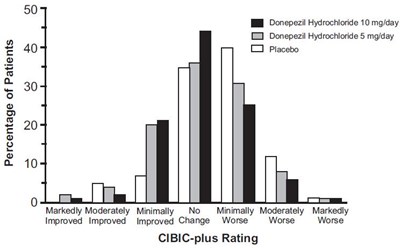
This is a chart representing the percentage of patients taking different dosages of Donepezil Hydrochloride and Placebo. The chart shows the CIBIC-plus Rating for each group of patients, ranging from Markedly Improved to Markedly Worse.*
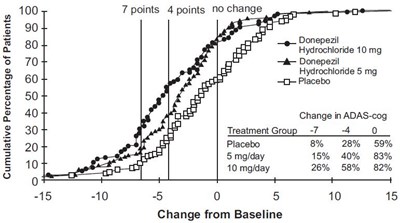
fig-5 - donepezil figure 5
This text appears to be a chart showing the change in ADAS-cog (a test used to measure cognitive function) for patients taking different doses of Donepezil Hydrochloride and a placebo. The chart shows the percentage of patients in each group who experienced a change of 7 points, 4 points or no change from their baseline score. The cumulative percentage of patients is also shown.*
fig-8 - donepezil figure 8
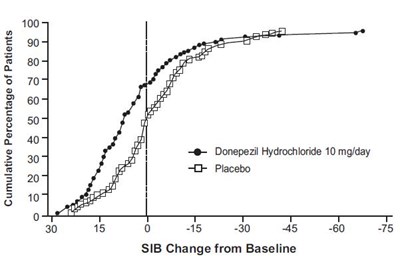
This is a graph that displays the cumulative percentage of patients and their scores on the SIB (Severe Impairment Battery) Change from Baseline scale for the medication Donepezil Hydrochloride at a dosage of 10mg/day versus a placebo. The x-axis represents timepoints from 0 to 75 weeks, while the y-axis represents the percentage of patients in each group who achieved or exceeded a certain SIB score change from baseline. However, no specific numbers are provided to indicate the actual percentage of patients at each timepoint or score, making it difficult to draw any concrete conclusions.*
fig-9 - donepezil figure 9

This appears to be a graph or chart showing the change in ADCS-ADL scores from the baseline for patients taking Donepezil Hydrochloride 10 mg/day versus a placebo over a period of 3-6 months of drug treatment. It also appears to indicate clinical improvement.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.