Product Images Clopidogrel
View Photos of Packaging, Labels & Appearance
- clopidogrel-100s - clopidogrel 100s
- clopidogrel-30s - clopidogrel 30s
- clopidogrel-500s - clopidogrel 500s
- clopidogrel-90s - clopidogrel 90s
- figure-03 - clopidogrel fig 3
- figure-1 - clopidogrel figure 01
- figure-2 - clopidogrel figure 02
- figure-4 - clopidogrel figure 04
- figure-5 - clopidogrel figure 05
- fig-7 - clopidogrel figure 07
- figure-3 - clopidogrel figure 3
- figure-6 - clopidogrel figure 6
- figure-8 - clopidogrel figure 8
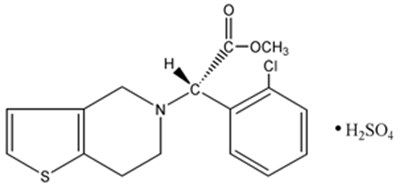
- structure - clopidogrel structure
Product Label Images
The following 14 images provide visual information about the product associated with Clopidogrel NDC 33342-060 by Macleods Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
clopidogrel-100s - clopidogrel 100s

Clopidogrel is a pharmaceutical drug that is available in the form of 75mg tablets manufactured by Madeods Pharmaceuticals Lid in Baddi, Himachal Pradesh, India. The batch number of this tablet, in particular, is HP/152/073342006012. The pharmacist is required to dispense a medication guide to every patient. The recommended dosage and storage for these tablets are also provided. However, the given text does not provide any other significant pieces of information.*
clopidogrel-30s - clopidogrel 30s

Clopidogrel is a medication presented in a bottle of 30 tablets of 75 mg each. Each tablet contains 97.875 mg of clopidogrel bisulfate, USP, which is equivalent to 75 mg of clopidogrel base. It is recommended to dispense the enclosed medication guide to each patient. The usual dosage areas see package insert. This medication should be stored at 20° to 25°C (68° to 77° F), although excursions permitted to 15° to 30° C (59° to 86° F) [see USP Controlled room temperature]. It is manufactured by Macleods Pharmaceuticals Ltd. for Macleods Pharma USA, Inc. The batch number is not available.*
clopidogrel-500s - clopidogrel 500s

Each Cleody tablet contains 75 mg of clopidogrel bisulfate, USP equivalent to 75 mg of clopidogrel base. The USP Controlled Room Temperature for storing is 20°C to 25°C (68°F to 77°F) with permitted excursions to 15°C to 30°C (59°F to 86°F). The usual dosage is defined in the package insert. Pharmacists are instructed to dispense the enclosed Medication Guide to patients. This medication is manufactured by Macleods Pharmaceuticals Ltd. with a code no. of HP/152/07, and is distributed by Macleods Pharma USA, Inc. in Plainsboro, NJ. This batch of Cleody has a shelf life of expiring on 30 X 23 mm.*
figure-2 - clopidogrel figure 02
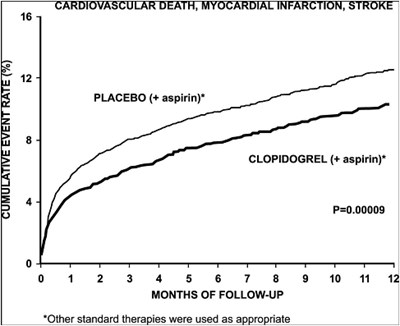
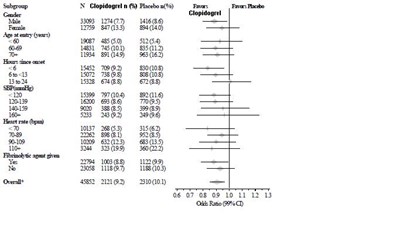
This is a comparison of two medications: Placebo plus aspirin and Clopidogrel plus aspirin. There is a numerical value given which is likely a statistical measurement of effectiveness or side effects. There is also a graph showing data over a 12 month period. Other standard therapies were used in conjunction with these medications.*
figure-4 - clopidogrel figure 04
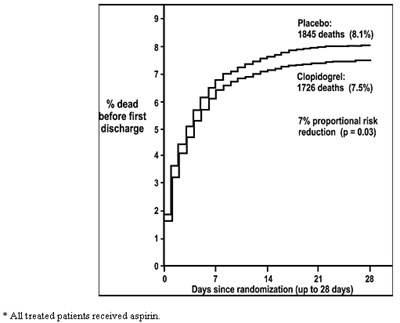
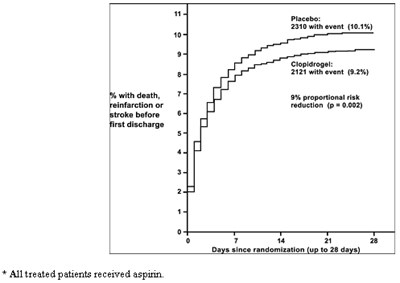
This is a description of a study evaluating the effectiveness of Clopidogrel compared to a placebo. The study involved patients who received aspirin and were followed up to 28 days after randomization. Results show a 7% reduction in proportional risk of death for patients taking Clopidogrel compared to placebo, with 1726 deaths (7.5%) in the Clopidogrel group and 1845 deaths (8.1%) in the placebo group.*
fig-7 - clopidogrel figure 07
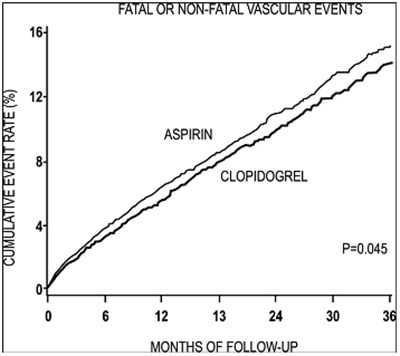
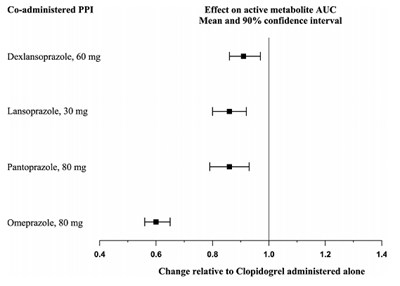
This text does not provide enough context to generate a useful description. It appears to be a table or graph showing data related to the drugs aspirin and clopidogrel, with a statistic of P=0.045 and a measure of months of follow-up. However, without more information, it is not possible to provide a meaningful interpretation.*
figure-3 - clopidogrel figure 3
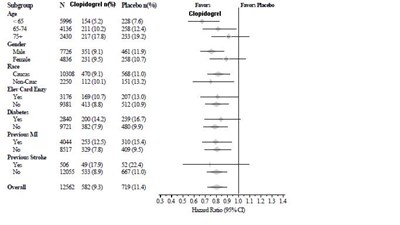
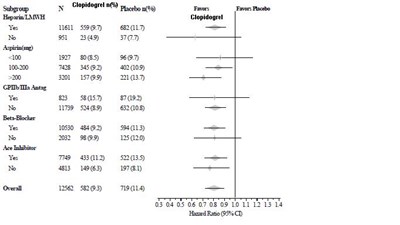
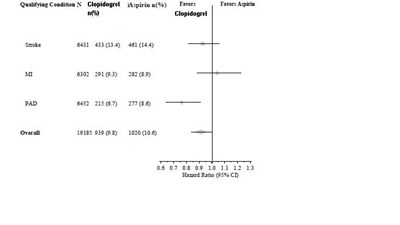
This is a table depicting numerical data related to the effectiveness of a medication called Clopidogrel. The table contains various code numbers and percentages indicating the success rate of the medication compared to a placebo. The text also includes a reference to a place called Ratho G5.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.