Product Images Rizatriptan
View Photos of Packaging, Labels & Appearance
- rizatriptan 10mg bottle 200s pack
- rizatriptan 10mg bottle 30s pack
- rizatriptan 5 mg 18s 3x6s
- rizatriptan 5 mg 18s 6x3s
- rizatriptan 5mg bottle 200s pack
- rizatriptan 5mg bottle 30s pack
- rizatriptan fct 10 mg 12s 2x6s
- rizatriptan fct 10 mg 12s 4x3s
- rizatriptan fct 10 mg 18s 3x6s
- rizatriptan fct 10 mg 18s 6x3s
- rizatriptan fct 5 mg 12s 2x6s
- rizatriptan fct 5 mg 12s 4x3s
- rizatriptan fct figure 5
- rizatriptan figure one
- rizatriptan figure two
- rizatriptan structure one
Product Label Images
The following 16 images provide visual information about the product associated with Rizatriptan NDC 33342-088 by Macleods Pharmaceuticals Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
rizatriptan 10mg bottle 200s pack

Rizatriptan Benzoate is a medication used in adults. The recommended dose for each tablet is 14.53 mg equivalent to 10 mg Rizatriptan Benzoate USP. This prescription drug should be stored at 20° to 25°C or 68° to 77° F. It can be stored at regulated temperature in the range of 15°C to 30°C. Macleods Pharma USA, Inc., has manufactured this particular batch of 200 tablets at Baddi in Himachal Pradesh, India. No additional information is provided.*
rizatriptan 10mg bottle 30s pack

MACLEOD's Rizatriptan Benzoate USP is a medication that comes in the form of tablets, containing 14.53mg and equivalent to 10mg Rizatriptan. The usual adult dose is available in the prescription information. It should be stored at a temperature of 20-25°C and the pharmacist should dispense it. It has been manufactured by Macleods Pharma USA, Inc, and Macleods Pharmaceuticals Ltd. The code number is HP/152/07 PMO3719101.*
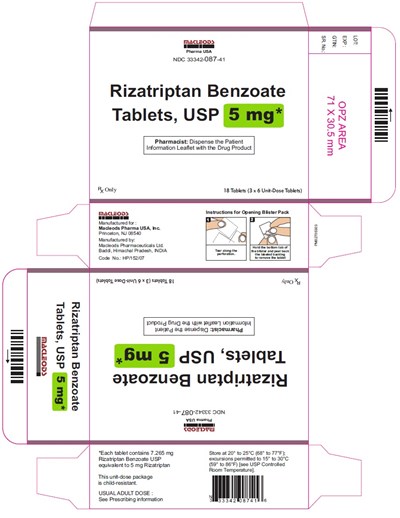
rizatriptan 5 mg 18s 6x3s

This is a packaging for Rizatriptan Benzoate Tablets, USP (5 mg). It contains 18 Tablets. The packaging includes instructions for opening the blister pack and information about the manufacturer. It also includes a warning to store the tablets at 20° to 25°C (68° to 77°F). The text contains some unreadable characters.*
rizatriptan 5mg bottle 200s pack

This is a description of a drug called Rizatriptan Benzoate available in tablet form. The tablets are 5mg each and the package includes 200 of them. The usual adult dose is described in the prescribing information included with the product. The tablets should be stored between 20° to 25°C (68° to 77° F) with an excursion permitted to 15°C to 30°C (59 to 86° F). The manufacturer is Macleods Pharma USA, Inc. with an address in Princeton, NJ.*
rizatriptan 5mg bottle 30s pack

Macleods Pharma USA manufactures Rizatriptan Benzoate Tablets with 5mg strength. This drug is dispensed with a patient information leaflet. Each tablet contains 7.265mg Rizatriptan Benzoate USP, equivalent to 5mg Rizatriptan. It is suggested to check the prescribing information for the usual adult dose. Store it between 20° to 25°C with permission to have an excursion to 15°C to 30°C. The tablet is imprinted with "MACLEODY" and "OPZ AREA 43 X 15 mm". The code number is HP/152107.*
rizatriptan fct 10 mg 12s 2x6s

This is a description of Rizatriptan Benzoate Tablets, USP manufactured by Macleods Pharma USA. The tablets contain 14.53mg equivalent to 10mg Rizatriptan Benzoate and are used for treatment of migraine headaches. The drug comes in a child-resistant unit-dose packaging, with each box containing 12 tablets. The recommended adult dose of the drug is available in the prescribing information.*
rizatriptan fct 10 mg 12s 4x3s

The text is not readable as it contains non-English characters and random characters.*
rizatriptan fct 10 mg 18s 3x6s

This is a packaging containing Rizatriptan Benzoate Tablets, USP. The package includes 18 tablets, which are divided into 3 unit-dose tablets. The manufacturer of the medicine is Macioods Pharma USA, and it is distributed with a patient information leaflet. The package provides instructions for opening the blister pack, and it is recommended to store the medicine at room temperature. The usual adult dose is not mentioned.*
rizatriptan fct 10 mg 18s 6x3s

This is a description of a medication called Rizatriptan Benzoate tablet made by Macleods Pharma USA. It comes in a 6x3 unit-dose pack and each tablet contains 1453mg of the active ingredient that is equivalent to 10mg of Rizatriptan. The tablets are used to treat migraines. The package has instructions for opening the child-resistant blister pack, and the usual adult dose can be found in the prescribing information. It should be stored at 20° to 25°C (68° to 77°F) with excursions permitted to 15° to 30°C (59° to 86°F) [see USP Controlled Room Temperature].*
rizatriptan fct 5 mg 12s 2x6s
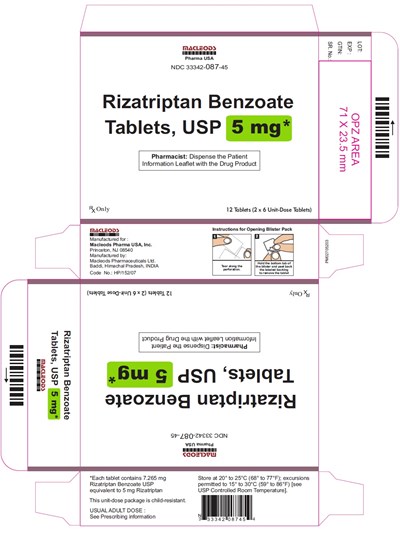
This is a description of a medication blister pack for Rizatriptan Benzoate tablets, USP with instructions for opening the pack. The pack contains 12 tablets in 2 x 6 unit-dose tablets and the pharmacist is instructed to dispense the patient information leaflet with the drug product. The pack is manufactured by Macleods Pharma USA, Inc., and contains tablets produced by Macleods Pharmaceuticals L1d. The tablets contain 7.265 mg of Rizatriptan Benzoate USP and are stored at a temperature range of 20°C to 25°C. The package is child-resistant and the usual adult dose should follow prescribing information.*
rizatriptan fct 5 mg 12s 4x3s

NDC 33342-087-50 is a package of Rizatriptan Benzoate tablets manufactured by Macleods Pharmaceuticals in India. Each tablet contains 5mg of the active ingredient and is designed to be a single-unit dose. The package includes instructions for opening the blister pack and dosage information as specified in the prescribing information. The storage conditions are specified as to keep the tablets at 20-25°C, with excursions allowed up to 15-30°C.*
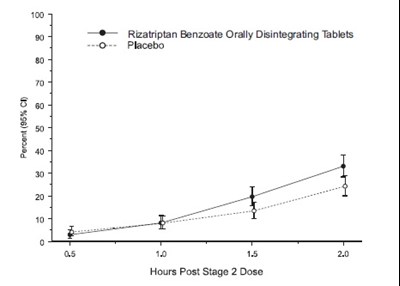
rizatriptan figure two
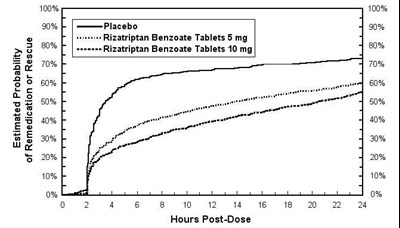
This appears to be a chart showing the estimated probability of remedication or rescue with different medications at different times post-dose. The medications listed are Placebo, Rizatriptan Benzoate Tablets 5 mg, and Rizatriptan Benzoate Tablets 10 mg. It also shows percentages ranging from 0% to 100%.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.