Product Images Losartan Potassium
View Photos of Packaging, Labels & Appearance
- Image of 100mg Label - LosartanPotassium100mgLabelTorrent
- Image of 25mg Label - LosartanPotassium25mgLabelTorrent
- Image of 50mg Label - LosartanPotassium50mgLabelTorrent
- Structural Formula - losartanpotassiumusp figure 01
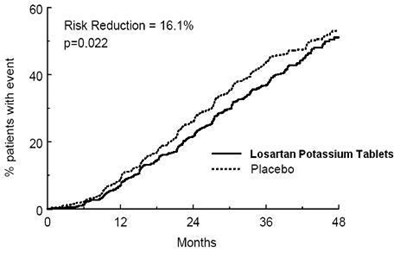
- FIGURE 1 - losartanpotassiumusp figure 02
- FIGURE 2 - losartanpotassiumusp figure 03
- FIGURE 3 - losartanpotassiumusp figure 04
- FIGURE 4 - losartanpotassiumusp figure 05
Product Label Images
The following 8 images provide visual information about the product associated with Losartan Potassium NDC 35356-949 by Lake Erie Medical Dba Quality Care Products Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Image of 100mg Label - LosartanPotassium100mgLabelTorrent

This is a description of a medication called Losartan Potassium in tablet form, packaged with 30 tablets per container. It is meant for oral consumption and each tablet contains 100 mg of Losartan Potassium. The medication should be stored in a controlled temperature between 68-77 degrees Fahrenheit and should be kept out of reach of children. It is cautioned not to be taken with alcohol without consulting a physician. The tablets are white to off-white colored, oval-shaped, biconvex, and film-coated with the number "100" on one side and "115" on the other side. The manufacturer is Torrent Pharms, Ltd located in India, and the NDC number is 35356-0883-30. Dosage information is provided either by consulting with a physician or by referring to the insert.*
Image of 25mg Label - LosartanPotassium25mgLabelTorrent

Losartan Potassium tablets are contained in a bottle with a child-resistant cap, and intended to be stored at a temperature between 68-77 degrees F. It is manufactured by Torrent Pharmaceuticals in Bharuch, India. Each oval-shaped tablet is film-coated, and contains 25 mg of Losartan Potassium. The recommended dosage should be consulted with a physician prior to taking. It is important to note that this medication should not be taken with alcohol. The tablets are identified by a code imprint of "25" on one side, and "113" on the reverse. The NDC for the bottle is 35356-0949-30.*
Image of 50mg Label - LosartanPotassium50mgLabelTorrent

This is a description of a medication called Losartan Potassium 50 mg. It comes in a container that is resistant to light and should be stored at a controlled temperature of 68-77 degrees F. It contains 30 tablets and each tablet has 50 mg of the active ingredient, Losartan Potassium. The medication is manufactured by Torrent Pharmaceuticals in India. There are warnings to keep it out of children's reach and to consult a physician for proper dosage.*
FIGURE 1 - losartanpotassiumusp figure 02
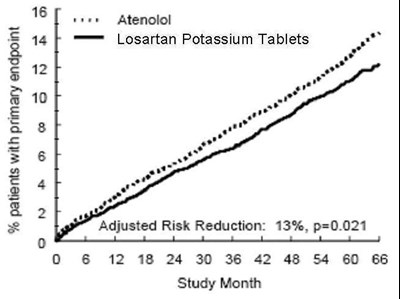
This is a clinical trial result summary showing the percentage of patients with the primary endpoint using Attenolol and Losartan Potassium Tablets. The adjusted risk reduction is 13% with a p-value of 0.021. The study duration was 60 months.*
FIGURE 2 - losartanpotassiumusp figure 03
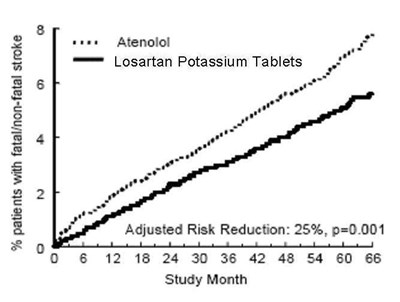
The text is describing the percentage of patients who had a fatal/non-fatal stroke and the efficacy of two medications - Atenolol and Losartan Potassium Tablets. The medication Losartan Potassium Tablets showed a 25% reduction in risk for stroke compared to Atenolol. The graph shows the study's timeline, from month 6 to month 66, and the change in risk over time.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.