FDA Label for Topcare Calamine
View Indications, Usage & Precautions
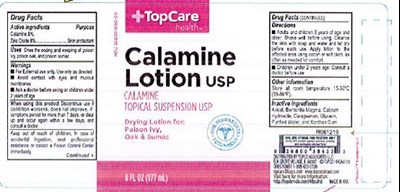
Topcare Calamine Product Label
The following document was submitted to the FDA by the labeler of this product Topco Associates Llc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Calamine 8% and Zinc Oxide 8%
Otc - Purpose
Skin protectant
Indications & Usage
dries the oozing and weeping o poison ivy, poison oak, and poison sumac.
Warnings
For external use only. Use only as directed.
Avoid contact with eyes and mucous membranes.
Ask a doctor before using on chilren 6 months of age.
Otc - Keep Out Of Reach Of Children
In case of accidental ingestion, seek professional assistance or contact a Poison Control center immediately.
Dosage & Administration
Adults and chidren 2 years of age and older: shake well before using. Cleanse the skin with soap and water and let it dry befroe each use. Apply lotion to the affected area using a cotton or soft cloth, as often as needed for comfort.
Children under 6 months of age: Consult a doctor before use.
Inactive Ingredient
Bentonite magma, calcium hydroxide, glycerin, purified water.
Package Label.Principal Display Panel
* Please review the disclaimer below.