FDA Label for Anti-itch
View Indications, Usage & Precautions
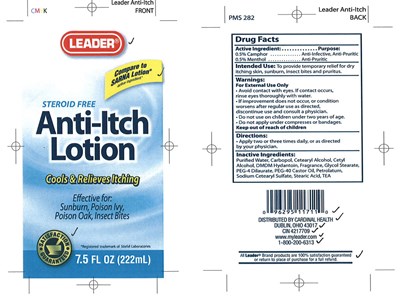
Anti-itch Product Label
The following document was submitted to the FDA by the labeler of this product Cardinal Health. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Drug Facts
Active Ingredient Purpose
0.5% Camphor Anti-Infective, Anti-Pruritic
0.5% Menthol Anti-Pruritic
Intended Use
To provide temporary relief for dry
itching skin, sunburn, insect bites and pruritus
Directions
Apply two or three times daily, or as directed by your physician
Warnings
Avoid contact with eyes.If contact occurs, rinse eyes thoroughly with water.
If improvement does not occur, or condition worsens after regular use as directed,
discontinue use and consult a physician.
Do not use on children under two years of age.
Do not apply under compresses or bandages
Inactive Ingredients
Purified water, Carbopol, Cetearyl alcohol, Cetyl alcohol, DMDM hydantoin,
Fragrance, Glycol stearate, PEG-4 Dilaurate, PEG-40 Castor oil, Petrolatum,
Sodium cetearyl sulfate, Stearic acid, TEA
Otc - Keep Out Of Reach Of Children
Keep out of reach of children
Dosage & Administration
Apply two or three times daily
Package Label.Principal Display Panel
* Please review the disclaimer below.