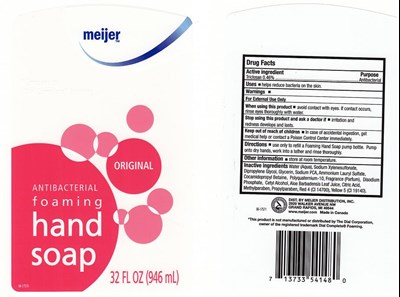
FDA Label for Antibacterial Foaming Original
View Indications, Usage & Precautions
Antibacterial Foaming Original Product Label
The following document was submitted to the FDA by the labeler of this product Meijer Distribution Inc. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient
TRICLOSAN 0.46%
Purpose
ANTIBACTERIAL
Uses
HELPS REDUCES BACTERIA ON THE SKIN
Warnings
FOR EXTERNAL USE ONLY
When Using This Product
AVOID CONTACT WITH EYES. IF CONTACT OCCURS, RINSE THOROUGHLY WITH WATER.
Stop Using This Product And Ask Doctor
IF IRRITATION AND REDNESS DEVELOP AND LAST.
Keep Out Of Reach Of Children
IN CASE OF ACCIDENTAL INGESTION, GET MEDICAL HELP OR CONTACT POISON CONTROL CENTER IMMEDIATELY.
Directions
PUMP ONTO DRY HANDS. WORK INTO A LATHER AND RINSE THOROUGHLY.
Other Information
STORE AT ROOM TEMPERATURE.
Inactive Ingredients
WATER (AQUA), SODIUM XYLENESULFONATE, DIPROPYLENE GLYCOL, GLYCERIN, SODIUM PCA, AMMONIUM LAURYL SULFATE, COCAMIDOPROPYL BETAINE, POLYQUATERNIUM-10, FRAGRANCE (PARFUM), DISODIUM PHOSPHATE, CETYL ALCOHOL, ALOE BARBADENSIS LEAF JUICE, CITRIC ACID, METHYLPARABEN, PROPYLPARABEN, RED 4 (CI 14700), YELLOW 5 (CI 19140).
Label Copy
* Please review the disclaimer below.