Product Images Lacosamide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Lacosamide NDC 42291-861 by Avkare, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
60 - 860 60

This is a medication description for NDC 42291-860-60 containing 50 mg of lacosamide, with dosage information available in the package insert. The tablets should be stored between 20-25°C (68-77°F) with excursions up to 25°C (86°F). The medication guide should be provided to each patient receiving the Rx. The tablets are manufactured by Amneal Pharmaceuticals and the label includes the manufacturer's address.*
60 - 861 60

NDC 42291-861-60 is a medication guide for Lacosamide tablets, USP, produced by Akare. Each film-coated tablet contains 100mg of lacosamide. It is advised to keep the tablets at a temperature of 20 to 25°C. The dosage information can be found in the package insert. Children should not have access to this medication. It is manufactured by Amneal Pharmaceutical of New York, LLC in Brookhaven.*
60 - 862 60

NDC 42291-862-60 is a medication called Lacosamide manufactured by AVKARE. It is a film-coated tablet that contains 150 mg of lacosamide, USP. The package insert contains dosage information. The medication should be stored at 20" to 25°C (66" to 77°F); excursions permitted between 1510 30°C (59" to 86'F) [see USP Controlled Room Temperature]. The medication is manufactured by Amneal Pharmaceuticals of New York, LLC, Brookhaven, NY 11719. The label warns to keep it out of the reach of children. There is also a non-varnished area for lot number, expiration date, and serialization.*
60 - 863 30

This is a medication information for Lacosamide, USP tablets. Each film-coated tablet contains 200mg of lacosamide. It comes in a bottle of 60 tablets and the recommended dosage is indicated in the package insert. The medication guide must be dispensed with each prescription, and the medication should be stored at the recommended temperature. It has been manufactured by Amneal Pharmaceuticals of New York, LLC and distributed by Avkare. The manufacturing and expiration date, lot number, and serialization are not available.*
2 - lacosamide tablets 2
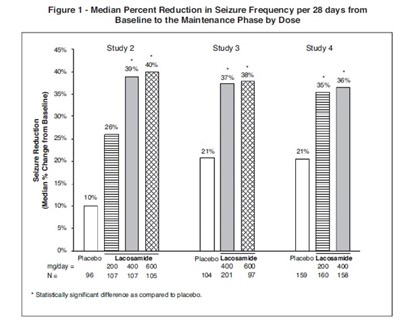
The text describes a figure (Figure 1) showing the percentage reduction in seizure frequency per 28 days during the Maintenance Phase by Dose. The data is presented for various medications, including Lacosamide and Placebo, at different mg/day doses. The figure indicates a statistically significant difference in seizure reduction in some of the treatments compared to Placebo.*
1 - lacosamide tablets 3
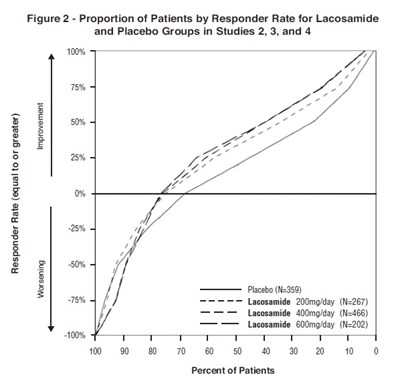
This is a graph labeled as "Figure 2" showing the proportion of patients by Responder Rate for the Lacosamide and Placebo groups in Studies 2, 3, and 4. The Responder Rate is expressed as a percentage on the y-axis, ranging from 0% to 100%. The x-axis shows the different levels of Responder Rate in increments of 10%. The graph displays three groups: Placebo (composed of 359 participants), Lacosamide 400mg/day (composed of 466 participants), and Lacosamide 600mg/day (composed of 202 participants).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
