Product Images Metformin Hydrochloride
View Photos of Packaging, Labels & Appearance
- PRINCIPAL DISPLAY PANEL - 500 mg - Container Label (90's count) - metformin hcl fig1
- PRINCIPAL DISPLAY PANEL - 500 mg - Blister Carton - 100 (10x10) Unit-Dose Tablets - metformin hcl fig2
- PRINCIPAL DISPLAY PANEL - 500 mg - Blister (1 x 10's count) - metformin hcl fig3
- PRINCIPAL DISPLAY PANEL - 850 mg - Container Label (90's count) - metformin hcl fig4
- PRINCIPAL DISPLAY PANEL -850 mg - Blister Carton - 100 (10 x 10) Unit-Dose Tablets - metformin hcl fig5
- PRINCIPAL DISPLAY PANEL - 850 mg - Blister (1 x 10's count) - metformin hcl fig6
- PRINCIPAL DISPLAY PANEL - 1000 mg - Container Label (60's count) - metformin hcl fig7
- PRINCIPAL DISPLAY PANEL - 1,000 mg - Blister Carton - 100 (10 x 10) Unit-Dose Tablets - metformin hcl fig8
- PRINCIPAL DISPLAY PANEL - 1,000 mg - Blister (1 x 10's count) - metformin hcl fig9
- Structure - metformin structure
Product Label Images
The following 10 images provide visual information about the product associated with Metformin Hydrochloride NDC 42385-947 by Laurus Labs Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 500 mg - Container Label (90's count) - metformin hcl fig1

Each tablet of Metformin Hydrochloride USP 500 mg, manufactured by Laurus Generics Inc. and packed in a light-resistant container, is intended for oral administration. The package insert contains information on dosage and administration. The drug is manufactured by Laurus Labs Limited in Visakhapatnam, India. It should be stored between 20° to 25°C (68° to 77°F) and excursions between 15° to 30°C (59° to 86°F) are permitted. USP 4 mentions the permissible temperature in controlled room temperature. The NDC number is 42385-947-90 and the drug is available in a pack of 90 tablets with the Rx-only label.*
PRINCIPAL DISPLAY PANEL - 500 mg - Blister Carton - 100 (10x10) Unit-Dose Tablets - metformin hcl fig2

This is a product description of Metformin Hydrochloride tablets containing 500 mg each. The text provides information about dosage, storage temperature, and the manufacturer's name and address.*
PRINCIPAL DISPLAY PANEL - 500 mg - Blister (1 x 10's count) - metformin hcl fig3

This appears to be a label of a medicine called "Metformin Hydrochloride", in the form of tablets with a strength of 500 mg. The label contains the drug's National Drug Code (NDC), manufacturing and expiration dates, lot numbers, and other related information. It is intended for prescription use only and should not be used without a doctor's advice.*
PRINCIPAL DISPLAY PANEL - 850 mg - Container Label (90's count) - metformin hcl fig4

Metformin Hydrochloride USP 850 mg film-coated tablets are a medication used to treat type 2 diabetes. The tablets should be stored between 20°C to 25°C (68°F to 77°F) but can be briefly kept between 15°C to 30°C (59°F to 86°F). Dispense the tablets in light-resistant containers. More information about dosage and administration can be found in the package insert. These tablets are manufactured by Laurus Labs Limited in Visakhapatnam, India and distributed by Laurus Generics Inc. in Berkeley Heights, NJ. The NDC number for this medication is 42385-948-90.*
PRINCIPAL DISPLAY PANEL -850 mg - Blister Carton - 100 (10 x 10) Unit-Dose Tablets - metformin hcl fig5

This is a description of a drug named "Metformin Hydrochloride Tablets, USP." The drug contains Vitormia Hyoctards and is used for dosage and Simnszion. The storage instructions indicate that the drug must be stored at a temperature between 2-25 degrees Celsius. The rest of the text is unreadable due to errors in .*
PRINCIPAL DISPLAY PANEL - 850 mg - Blister (1 x 10's count) - metformin hcl fig6

This is a description of a medication called Metformin Hydrochloride. It comes in tablet form and is only available with a prescription. The tablets are manufactured by Laurus Labs Limited and have a unique identification number (NDC) of 42385-948-10. The text also contains several product codes, lot numbers, and manufacturing information.*
PRINCIPAL DISPLAY PANEL - 1000 mg - Container Label (60's count) - metformin hcl fig7

Each tablet contains 1,000 mg of Metformin Hydrochloride, a medication used to treat type 2 diabetes. The tablets are film-coated and are packaged in bottles of 60. Dosage and administration information can be found in the package insert. The medication should be stored in light-resistant containers at a temperature of 20° to 25°C (68° to 77°F), with excursions permitted between 15° to 30°C (59° to 86°F). The manufacturer is listed as Laurus Labs, with an address in Visakhapatnam-531011, India. This medication is only available with a prescription.*
PRINCIPAL DISPLAY PANEL - 1,000 mg - Blister Carton - 100 (10 x 10) Unit-Dose Tablets - metformin hcl fig8
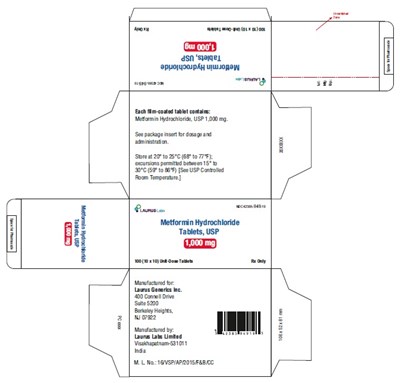
This is a product description of Metformin Hydrochloride Tablets of 1000mg strength, manufactured by a company named Launs Lbe Uitd. The product seems to be available for purchase at a price of $5.00 per pack. The text mentions some instructions regarding storage and usage of the drug, along with some codes and numbers like Sorea 2010 20 68 10 7%, ACET 1o 86 (S0 USPCanvoted) and Foan Tempertue. The description also includes an address of the manufacturer (Suas0 sty Haigts, W22 | Ventsared vy, Vietnam) and some unreadable text at the end.*
PRINCIPAL DISPLAY PANEL - 1,000 mg - Blister (1 x 10's count) - metformin hcl fig9

This is a label information for Rx-only Metformin Hydrochloride Tablet, USP 1,000 mg produced by Laurus Labs Limited. It includes the drug's NDC code, M.L. No., and manufacturer details. It also contains a lot and expiration date which are not readable due to errors.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
