Product Images Buspirone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Buspirone Hydrochloride NDC 42543-742 by Strides Pharma Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
5mg-dosage adjustment - b0825ce2 82b8 4850 9e90 4259728823b5 02
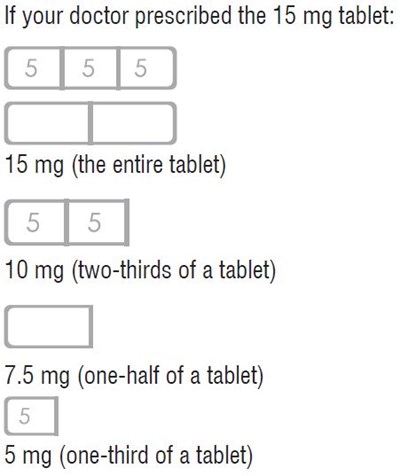
This is a dosage guide for a 15 mg tablet prescribed by a doctor. It provides information on how to split the tablet into smaller doses of 5 mg, 10 mg, 7.5 mg and 5mg.*
30-10mg-dose adjustment - b0825ce2 82b8 4850 9e90 4259728823b5 04
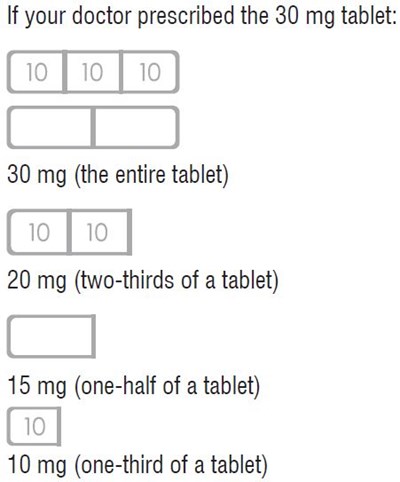
This text appears to be a set of dosing instructions for a 30 mg tablet of medicine. It provides information on what dosage to take based on how much of the tablet is consumed, ranging from the whole tablet (30 mg) to one-third of the tablet (10 mg).*
5mg-500s - b0825ce2 82b8 4850 9e90 4259728823b5 06

Each tablet contains 5 mg of Buspirone Hydrochloride, USP. The manufacturer is located in India and the product is distributed by Strides Pharma in East Brunswick, NJ. The tablets should be stored at controlled room temperature. The package should be tight, light-resistant and have a child-resistant closure. No other information is provided.*
7.5mg-100s - b0825ce2 82b8 4850 9e90 4259728823b5 07

Each tablet of this medication contains 7.5 mg of Buspirone Hydrochloride. The usual dosage is BusPlROfle. The medication should be kept in a tightly closed container and away from children. Store it in a cool and dry place at a temperature range of 20 °C to 25 °C (68 °F to 77 °F), and excursions are permitted between 15 °C to 30 °C (59 °F to 86 °F). The manufacturer is Strides Pharma Science Ltd, and it is distributed by Strides Pharma Inc. No additional information is available.*
10mg-500s - b0825ce2 82b8 4850 9e90 4259728823b5 08

Each tablet contains 10 mg of Buspirone Hydrochloride, USP. Dosage information is available in the package insert. Store in a container with a child-resistant closure at controlled room temperature. This product is manufactured by Strides Pharma Science Ltd. and distributed by Strides Pharma Inc. in East Brunswick, NJ. This is a prescription-only medication with 500 tablets per container. Batch details are printed on the packaging.*
15mg-180s - b0825ce2 82b8 4850 9e90 4259728823b5 09

Each tablet of Buspirone Hydrochloride, USP contains 15mg of the active ingredient. The tablets are manufactured by Strides Pharma Science Ltd. in Bengaluru, India, and distributed by Strides Pharma Inc. in the US. The usual dosage instructions are not available. The tablets should be stored between 20°C-25°C (68°F-86°F) and in a light-resistant container. The lot number is provided for batch details. The code number is KRIDRUGS/KTK/25/415/98 and the tablets are sold in a bottle with 180 tablets. This is a prescription drug.*
30mg-60s - b0825ce2 82b8 4850 9e90 4259728823b5 10

This is a drug label for Buspirone Hydrochloride manufactured by Strides Pharma Science Ltd. The label specifies the usual dosage, storage requirements, and batch details for the drug. It is available in tablet form and is only available on prescription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.


