Product Images Midazolam
View Photos of Packaging, Labels & Appearance
- midazolam-crtna.jpg - midazolam crtna
- midazolam-crtnb.jpg - midazolam crtnb
- midazolam-crtnc.jpg - midazolam crtnc
- midazolam-lbla.jpg - midazolam lbla
- midazolam-lblb.jpg - midazolam lblb
- midazolam-lblc.jpg - midazolam lblc
- midazolam-stra.jpg - midazolam stra
- midazolam-strb.jpg - midazolam strb
- midazolam-strc.jpg - midazolam strc
Product Label Images
The following 9 images provide visual information about the product associated with Midazolam NDC 42571-414 by Micro Labs Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
midazolam-lblc.jpg - midazolam lblc

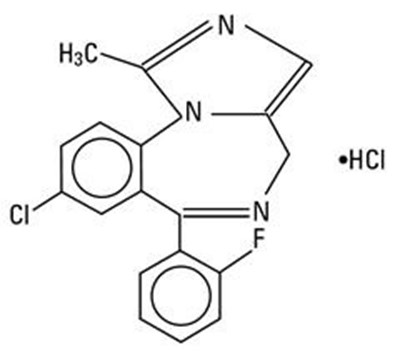
Midazolam Injection, USP is a medication indicated for intravenous or intramuscular use. It is available in a 2 mL single-dose vial with a strength of 10 mg/2 mL (5 mg/mL). The medication is manufactured by Sterile Micro Labs USA, Inc. in Somerset, NJ. The vial is preservative-free and any unused portion should be discarded. The product is made in India and the expiration date is March 2023.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.