Product Images Gabapentin
View Photos of Packaging, Labels & Appearance
- Cockcroft and Gault Equation - image 04
- Gabapentin Structural Formula - image 05
- Figure 1. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 1 - image 06
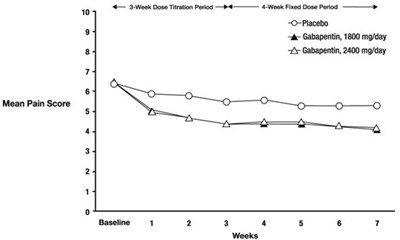
- Figure 2. Weekly Mean Pain Scores (Observed Cases in ITT Population): Study 2 - image 07
- Figure 3. Proportion of Responders (patients with ≥ 50% reduction in pain score) at Endpoint: Controlled PHN Studies - image 08
- Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjuctive Therapy Studies in Patients Greater than or Equal to 12 Years of Age with Partial Seizures - image 09
- image description - label 100mg
- image description - label 300mg
- image description - label 400mg
Product Label Images
The following 9 images provide visual information about the product associated with Gabapentin NDC 42582-115 by Bi-coastal Pharma International Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Cockcroft and Gault Equation - image 04
This appears to be a formula for calculating creatinine clearance (CLCr) using the patient's age and weight. However, some of the characters in the formula are not readable due to errors in the . Without further context, it is not clear what T3y and the unit ugdl represent. The formula also notes that female patients have a different factor (85) than male patients for calculating CLCr.*
Figure 3. Proportion of Responders (patients with ≥ 50% reduction in pain score) at Endpoint: Controlled PHN Studies - image 08
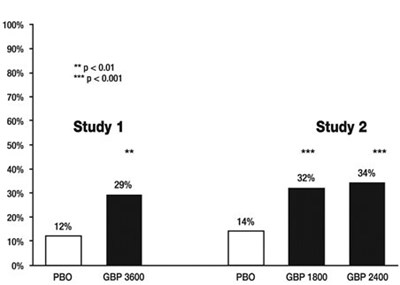
Figure 4. Responder Rate in Patients Receiving Gabapentin Expressed as a Difference from Placebo by Dose and Study: Adjuctive Therapy Studies in Patients Greater than or Equal to 12 Years of Age with Partial Seizures - image 09
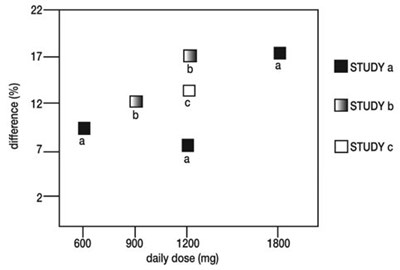
image description - label 100mg

This is a description of Gabapentin Capsules, USP distributed by Bi-Coastal Pharma International LLC. Each capsule contains 100 mg of Gabapentin, USP. The pharmacist is advised to dispense the medication guide separately to each patient. The prescription should be stored between 20-25°C (68-77°F) with excursions permitted between 15°-30°C (59°-86°F). Dispense in a well-closed container as defined in the USP. The package insert should be reviewed for full prescribing information. It was manufactured in Goa, India by Marksans Pharma Ltd. The lot number and expiration date are not available.*
image description - label 300mg

NDC 42582-115-10 is a medication called Gabapentin manufactured by Marksans Pharma in India and distributed by Bi-Coastal Pharma International LLC . It is available in the form of capsules, USP containing 300 mg Gabapentin in each capsule. Dispensing of this medication must be accompanied by a medication guide provided to each patient. For dosage and use, the package insert should be thoroughly read. It should be stored at 20° to 25°C (68° to 77°F) with excursions allowed between 15° - 30°C (59° - 86°F) [See USP Controlled Room Temperature]. This medication should be dispensed in a well-closed container as defined in the USP. The lot number and expiry date are also provided.*
image description - label 400mg

This is a description of Gabapentin Capsules, USP medication manufactured by Marksans Pharma Ltd. and distributed by Bi-Coastal Pharma International LLC. The medication comes in the form of 100 capsules, each containing 400mg of Gabapentin, USP. It is a prescription-only medication and comes with a Medication Guide. The dosage and usage guidelines are provided in the package insert. The medication should be stored at a temperature range of 20° to 25°C (68° to 77°F), with permitted excursions between 15° - 30°C (59° - 86°F). It should be dispensed in well-closed containers and has a unique LOT# and EXP. DATE.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.