Product Images Bisoprolol Fumarate And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
- 10mg 30 - 10 mg 30 count
- 10mg 500 - 10 mg 500 count
- 10mg 100ct
- 25 100 - 25 mg 100 count
- 25mg 30 - 25 mg 30 count
- 25 500 - 25 mg 500 count
- 5mg 100 - 5 mg 100 count
- 5mg 30 - 5 mg 30 count
- 5mg 500 - 5 mg 500 count
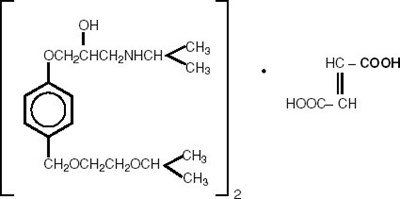
- BISO API Structure - api pic new1
- HCTZ API Structure - api pic new2
- Table 1 - table 1 new
- table-3 - table 3 new
- table-2 - table new 2
Product Label Images
The following 14 images provide visual information about the product associated with Bisoprolol Fumarate And Hydrochlorothiazide NDC 42799-922 by Edenbridge Pharmaceuticals Llc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10mg 500 - 10 mg 500 count

This is a medication called Bisoprolol Fumarate and Hydrochlorothiazide in tablet form with a strength of 10 mg and 6.25 mg respectively. This medicine is available in a bottle containing 500 tablets.*
25mg 30 - 25 mg 30 count

Bisoprolol Fumarate and Hydrochlorothiazide Tablets, USP are a medication used to treat high blood pressure. Each tablet contains 25mg of bisoprolol fumarate and 625mg of hydrochlorothiazide. The bottle contains 30 tablets. The text also includes some gibberish characters that may be a result of an error in the process.*
25 500 - 25 mg 500 count

This is a label for a bottle of Bisoprolol Fumarate and Hydrochlorothiazide Tablets, USP with a strength of 25mg/6.25mg and a quantity of 500 tablets.*
5mg 100 - 5 mg 100 count

The text contains the names of two drugs, Bisoprolol Fumarate and Hydrochlorothiazide, in tablet form with a dose of 5mg of Bisoprolol Fumarate and 625mg of Hydrochlorothiazide.*
5mg 30 - 5 mg 30 count

This is a prescription pharmaceutical with the name Bisoprolol Fumarate and Hydrochlorothiazide. It is available in tablet form with dosages of 5mg and 25mg.*
Table 1 - table 1 new
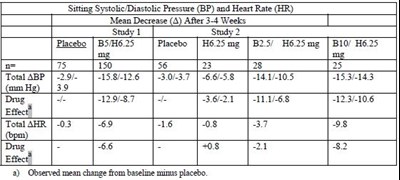
The text is reporting results of a study on Sitting Systolic/Diastolic Blood Pressure (BP) and Heart Rate (HR) reduction after 3.4 weeks of treatment with different drugs. The table presents the study results for Placebo, BSH6.25, H625mg, B3/H62smg, and BI0/H625 drugs in Study 1 and Study 2. The study reports the mean decrease (A) for BP and HR and the observed mean change from baseline minus placebo.*
table-3 - table 3 new

This is a table describing the adverse experiences of patients in a clinical trial. It lists various body systems and corresponding adverse experiences, such as chest pain and cough. The table also indicates the percentage of patients who experienced drug-related adverse effects.*
table-2 - table new 2

This appears to be a table containing data on serum potassium levels in a study conducted in the US. The study was likely placebo-controlled, but there is not enough information to determine the specifics. The table shows mean change in serum potassium levels measured in mEq/L between 2004 and 7011 for both the Placebo and the treatment group ("B10)"). It also indicates the percentage of participants who experienced hypokalemia (low potassium levels) in each group.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.