Product Images Topiramate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Topiramate NDC 43063-734 by Pd-rx Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
43063734 Label - 43063734
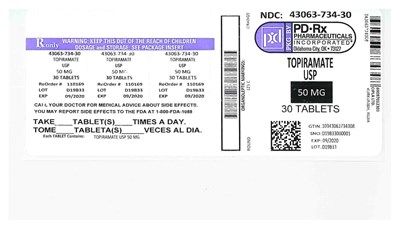
This text contains information related to the dosage and storage of Topiramate USP 50 mg tablets. The package contains 30 tablets, to be taken "tablet(s) times a day." The package insert and organoleptic markings are also included. The text also includes a warning to keep the medication out of reach of children and to contact a doctor for medical advice about side effects. The product has a GTIN of 10343063734308 and LOT D19B33, and must be used before 09/2020. It also provides a number to report side effects to the FDA.*
ccb7a1e4 a3a1 4a0e 8eb0 8086f53e314b 02
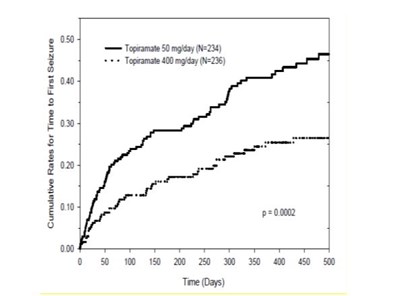
This appears to be a graph showing the cumulative rates for time to first seizure for two groups of individuals taking different doses of topiramate. The x-axis represents time in days and the y-axis represents the cumulative rate for time to first seizure. However, without more context or information, it is difficult to provide a more detailed description or analysis.*
ccb7a1e4 a3a1 4a0e 8eb0 8086f53e314b 03

During the double-blind phase, the average monthly migraine period rate increased by 56 percent from baseline using 100mg/day to 200mg/day TOPAMAX. The statistical analysis showed a significant difference (p<0.001) and (p<0.0001).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
