Product Images Ondansetron
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 3 images provide visual information about the product associated with Ondansetron NDC 43063-770 by Pd-rx Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
43063770 Label - 43063770

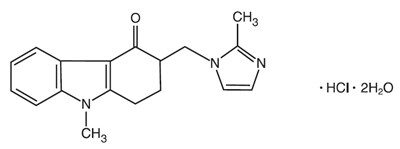
This is a medication warning label with instructions for dosage and storage of Ondansetron tablets. It also includes information on reporting side effects and contacting a doctor for medical advice. The tablets are oval and yellow with organoleptic markings of 8G. Each tablet contains 1mg of Ondansetron hydrochloride dihydrate equivalent to 8 mg of Ondansetron USP. It is meant to be taken multiple times a day. There is also a lot number, expiration date, and contact information for the manufacturer. The last few lines seem to be gibberish and cannot be evaluated.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.