Product Images Rocuronium Bromide
View Photos of Packaging, Labels & Appearance
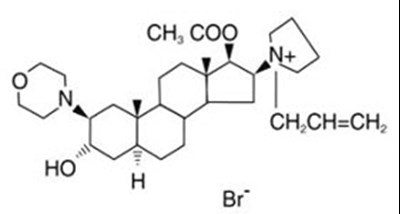
- Rocuronium Structural Formula - image 01
- Figure 1 - Rocuronium Function of Dose - image 02
- Figure 2 - Rocuronium Function of Dose - image 03
- Figure 3 - Rocuronium Clinical Durations - image 04
- Rocuronium Representative Container Label 43066-007-10 - image 05
- Rocuronium Representative Carton Label 43066-007-10 1 of 2 - image 06
- Rocuronium Representative Carton Label 43066-007-10 2 of 2 - image 07
- Rocuronium Representative Container Label 43066-013-10 - image 08
- Rocuronium Representative Carton Label 43066-013-10 1 of 2 - image 09
- Rocuronium Representative Carton Label 43066-013-10 2 of 2 - image 10
- image 11
- image 12
- image 13
- image 14
Product Label Images
The following 14 images provide visual information about the product associated with Rocuronium Bromide NDC 43066-013 by Baxter Healthcare Corporation, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Rocuronium Representative Container Label 43066-013-10 - image 08

This is a warning label for Rocuronium Bromide Injection, a paralyzing agent used for intravenous use only. No additional information is available.*
Rocuronium Representative Carton Label 43066-013-10 2 of 2 - image 10

NDC 43066-013-10 is a 10mL multiple dose vial containing Rocuronium Bromide injection, a paralyzing agent available for IV use only. It has a shelf life of 30 days and must be discarded afterwards. This product is manufactured by Bocer et Corporation.*
image 13

This is a medication called Rocuronium Bromide, which comes in the form of vials that contain 100mg of the active ingredient in 10mL of liquid (10mg/mL). It is administered through intravenous use only. However, it is essential to note the WARNING label that this medication is a paralyzing agent, and therefore, it should only be used by authorized healthcare professionals. The manufacturer is R oniy Baxter.*
image 14

Rocuronium Bromide Injection is a medication used during surgery to relax muscles and facilitate endotracheal intubation.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.