Product Images Losartan Potassium Tablets, 100 Mg
View Photos of Packaging, Labels & Appearance
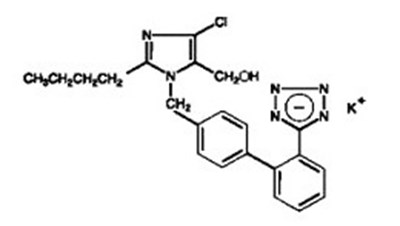
Product Label Images
The following 7 images provide visual information about the product associated with Losartan Potassium Tablets, 100 Mg NDC 43353-299 by Aphena Pharma Solutions - Tennessee, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - figure2

This appears to be a graph of a study comparing the effects of Atenolol and Losartan Potassium on patients with fatal or non-fatal stroke. The graph shows the adjusted risk reduction of 25%, with a p-value of 0.001. The study was conducted over a period of 66 months.*
Figure 3 - figure3

This appears to be a chart or graph displaying results of a study evaluating the effects of two medications, Losartan and Atenolol, on the incidence of stroke in different demographic subgroups. The primary endpoint was a composite of fatal and non-fatal stroke events. The chart shows the number of events and hazard ratios for each subgroup, including age, gender, race, diabetes, and history of cardiovascular disease. Symbols on the chart are proportional to sample size. There is not enough context to determine the overall results or conclusions of the study.*
Figure 4.jpg - figure4

This is a partial data table with some numerical values and drug names. The number 80 appears to be related to patients experiencing an "event." There is also a percentage value of "Risk Reduction = 16.1%". The drug names mentioned in the table are Losartan Potassium and Placebo. There are some numbers (022, 12, 24, 48) but it is unclear what they refer to without more context.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.