Product Images Azithromycin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Azithromycin NDC 43386-471 by Lupin Pharmaceuticals,inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
300 mg - 29ca4bfb 67cc 4531 8d9c 598a03e60fcc 02

NDC 43386-470-60 is a bottle of Azithromycin monohydrate powder for oral suspension used for treating bacterial infections in adults and children. The bottle contains 300mg of Azithromycin and can be used to make a 15mL suspension. The dosage and usage information can be found in the accompanying prescribing information. The dry powder should be stored at 20 to 25°C (68° to77°F) and protected from freezing. After mixing with water, the suspension can be stored at 5° to 30°C (41° to 86°F) and should be used within 10 days. The bottle should be tightly closed and kept out of the reach of children. The medicine has been manufactured by Novel Laboratories, Inc in Somerset, NJ 08873 and is distributed by Lupin Pharmaceuticals, Inc in Baltimore, MD 21202. The bottle has a cherry flavor.*
carton-300 mg - 29ca4bfb 67cc 4531 8d9c 598a03e60fcc 03
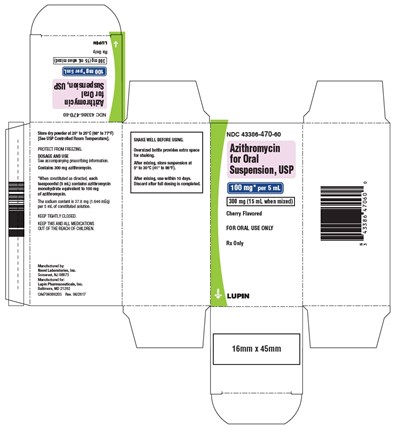
This is a medication or drug called Azithromycin presented in a bottle for oral suspension use. The bottle contains 300 mg and should be stored at room temperature and protected from freezing. The dosage and use of the medication should be consulted on the accompanying prescription form. The label presents the drug name, manufacturer's contact details, the prescription number, and the National Drug Code (NDC). There is also a caution note indicating that the drug is for oral use only.*
1200 mg- container - 29ca4bfb 67cc 4531 8d9c 598a03e60fcc 04

This text describes the usage and storage instructions of an oral suspension containing 1200mg azithromycin, used for treating various infections. The suspension is to be stored at a controlled room temperature and protected from freezing. The mixing directions require adding water and shaking the bottle well before use. The medicine is to be used within ten days of mixing and kept tightly closed in a dry place. The product is manufactured by Lupin Pharmaceuticals, Inc. and Novel Laboratories, Inc. and is only available via prescription. The text also confirms that the oral suspension is cherry-flavored.*
1200 mg- carton - 29ca4bfb 67cc 4531 8d9c 598a03e60fcc 05
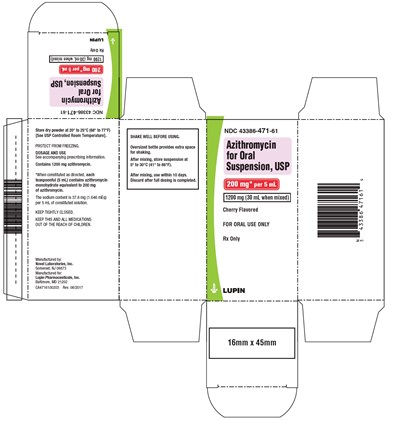
This is a medication label containing important information on dosage and use for Azithromycin oral suspension, USP- a drug used to treat bacterial infections. The text also provides information on how to store the medication and the manufacturer’s details. Users are advised to keep the medication out of the reach of children, store it within a recommended temperature range, shake well before use, and use within ten days after mixing.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.