Product Images Irbesartan And Hydrochlorothiazide
View Photos of Packaging, Labels & Appearance
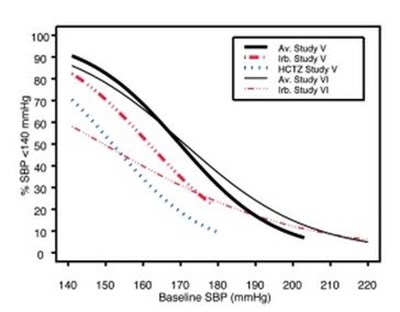
- Irbesartan and Hydrochlorothiazide Figure 1A - 22bf08c2 615b 4b48 af36 1b71a91459dc 01
- Irbesartan and Hydrochlorothiazide Figure 1B - 22bf08c2 615b 4b48 af36 1b71a91459dc 02
- Irbesartan and Hydrochlorothiazide Figure 2A - 22bf08c2 615b 4b48 af36 1b71a91459dc 03
- Irbesartan and Hydrochlorothiazide Figure 2B - 22bf08c2 615b 4b48 af36 1b71a91459dc 04
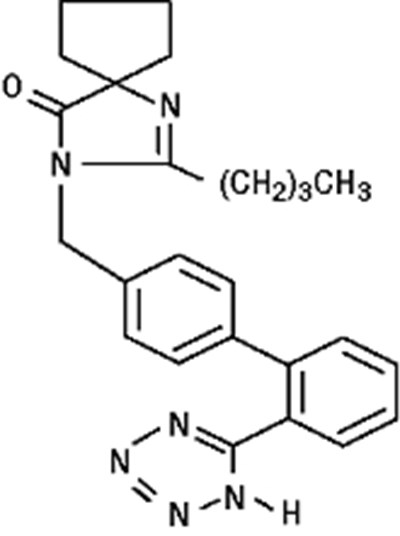
- Chemical Structure Irbesartan - 22bf08c2 615b 4b48 af36 1b71a91459dc 05
- Chemical Structure Hydrochlorothiazide - 22bf08c2 615b 4b48 af36 1b71a91459dc 06
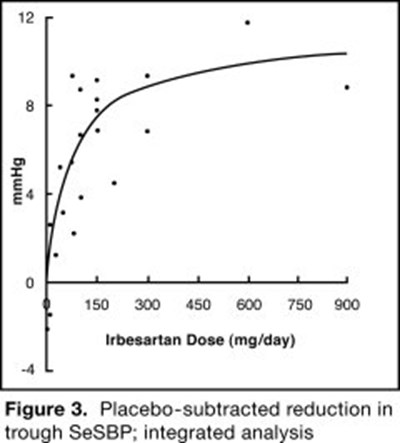
- Irbesartan and Hydrochlorothiazide Figure 3 - 22bf08c2 615b 4b48 af36 1b71a91459dc 07
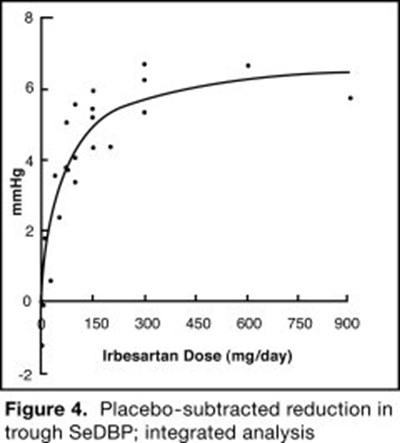
- Irbesartan and Hydrochlorothiazide Figure 4 - 22bf08c2 615b 4b48 af36 1b71a91459dc 08
- Irbesartan and Hydrochlorothiazide 150-12.5 mg - 22bf08c2 615b 4b48 af36 1b71a91459dc 09
- Irbesartan and Hydrochlorothiazide 300/12.5 mg - 22bf08c2 615b 4b48 af36 1b71a91459dc 10
Product Label Images
The following 10 images provide visual information about the product associated with Irbesartan And Hydrochlorothiazide NDC 43547-331 by Solco Healthcare Us, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Irbesartan and Hydrochlorothiazide 150-12.5 mg - 22bf08c2 615b 4b48 af36 1b71a91459dc 09

Each film-coated tablet contains 150mg of hesaanand and 125mg of Fyarochoothaze. The package contains dosing information. Store between 20°C and 25°C (68°F-77°F) with excursions permitted between 15°C and 30°C (59°F-86°F) (see USP Controlled Room Temperature). Manufactured by nesongHuohoi Pharmaceuticals, Yunaiao, Livs Zhefang 47024, China. Distributed by Soko Healthcare US, LLC, Somerset, NJ 08873, USA. The tablets are USP 150/12.5 mg, and this product is available only with a prescription. There is an FPO box on the package to indicate the area to be left uncoated.*
Irbesartan and Hydrochlorothiazide 300/12.5 mg - 22bf08c2 615b 4b48 af36 1b71a91459dc 10

Each film-coated tablet contains 300 mg of Valsartan and 125 mg of Hydrochlorothiazide. The NDC number is 43547:331.03 and it is available by prescription only. The storage temperature is between 20° and 25°C (68°F and 77°F) with excursions allowed from 15°C to 30°C (59°F to 86°F). The manufacturer is Zhejiang Huahai Pharmaceutical Co. Ltd, located in Kunshan City, Jiangsu, China, and it is distributed by Lek Pharmaceuticals, a subsidiary of Sandoz Inc. The drug name is "VG LT EPAT" and it is a USP tablet with a strength of 300/12.5 mg. This text appears to relate to a medication.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.