Product Images Paroxetine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Paroxetine NDC 43547-350 by Solco Healthcare Us, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
container label 10mg 30ct - 6cfde064 ddcb 49df a53c 953c40eb1470 02
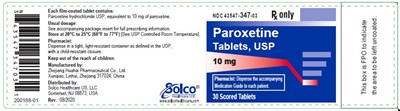
This is a medication label for Paracetamol tablets containing 10mg of Paracetamol Hydrochloride, intended for use as directed on the package insert. It is manufactured by Zhejang Hushai Pharmaceutical Co. in a child-resistant container and should be kept out of reach of children.*
container label 20mg 30ct - image 01

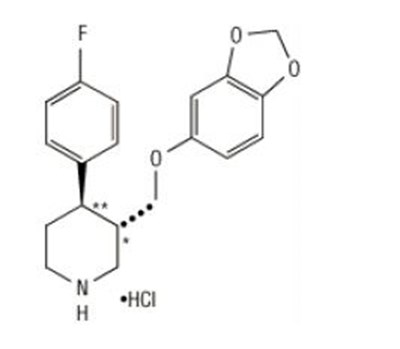
This is a description of Paroxetine Tablets, USP. Each film-coated tablet contains Paracetamol Hydrochloride that is equivalent to 20 mg of Paracetamol. It is important to store the tablets at a temperature range of 2°C to 25°C and in a light-resistant container with child-resistant closure. Complete prescription information can be found in the accompanying package insert. The manufacturer is Zhejiang Hushai Pharmaceutical Co. Ltd, China, and the distributor is Solo Healthcare US, LLC, Somerset, NJ. The tablets are dispensed by pharmacists and are sold in packages of 30 scored tablets.*
container label 30mg 30 ct - image 02

This is a description of a medication called Paroxetine. Each tablet contains Paroxetine hydrochloride 30 mg along with other inactive ingredients. The recommended dosage is not mentioned in the text and can be found in the package insert. The tablets should be stored at room temperature, and dispensed in a tight, light-resistant container with child-resistant closure for safety. The medication is manufactured by Zhejang Hushai Pharmaceutical Co. L located in China and distributed by a company named Sandoz. The last sentence seems to refer to the instructions for leaving an area of the tablet uncoated possibly for the purpose of scoring.*
CONTAINER LABEL 40mg 30ct - image 03

This appears to be a label for a medication called Paroxetine, containing film-coated tablets. The label provides information on dosage, prescribing information, manufacturer and distributor information, as well as instructions for storage. Additionally, the label includes a warning to keep the medication out of reach of children and a reference to a medication guide.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.