Product Images Losartan Potassium
View Photos of Packaging, Labels & Appearance
- 25 mg, 90 Tablets/bottle - 8ca0c9d3 3a50 4efe 92df 30a0c50d48a7 06
- 50 mg. 30 Tbalets/bottle - 8ca0c9d3 3a50 4efe 92df 30a0c50d48a7 07
- 100 mg, 30 Tablets/bottle - 8ca0c9d3 3a50 4efe 92df 30a0c50d48a7 08
- Chemical Structure - spl image1 structure1
- Figure 1 - spl image2 figure1
- Figure 2 - spl image3 figure2
- Figure 3 - spl image4 figure3
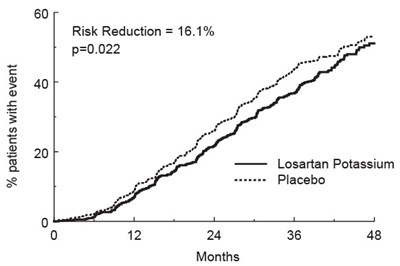
- Figure 4 - spl image5 figure4
Product Label Images
The following 8 images provide visual information about the product associated with Losartan Potassium NDC 43547-362 by Solco Healthcare Us, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
25 mg, 90 Tablets/bottle - 8ca0c9d3 3a50 4efe 92df 30a0c50d48a7 06

This is a description of a medication container. The medication is Losartan Potassium, which is in tablet form and comes in a container with a controlled room temperature of 20-25 degrees Celsius. The container was manufactured by Fong i PramasescalCo and distributed by Soeo st S. The container has a dispense-able cap for ease of use, indicated with "ROVIACET Peas dsperse". The dosage and usage instructions are not available.*
50 mg. 30 Tbalets/bottle - 8ca0c9d3 3a50 4efe 92df 30a0c50d48a7 07

This appears to be a description of a medication named Losartan Potassium, available in the US. It is manufactured by a company called Sandoz, and comes in a container with 28 tablets. The tablets should be kept in a tightly closed container at room temperature. The label also includes information about the medication's chemical composition and physical characteristics, such as its melting point and the fact that it is microcrystalline. It is not possible to interpret the purpose of the medication or any potential medical benefits from this information alone.*
Figure 1 - spl image2 figure1

This text appears to be a comparison between two medications, Atenolol and Losartan Potassium, in relation to the primary endpoint in a study that took place over 66 months. The adjusted risk reduction for Losartan Potassium was 13%, with a p-value of 0.021. The rest of the text is a timeline of the study months.*
Figure 2 - spl image3 figure2

This is a comparison of two medications, Atenolol and Losartan Potassium, in terms of their adjusted risk reduction for fatal/non-fatal stroke. The percentage of patients experiencing stroke is reported for different study months from 6 to 66.*
Figure 3 - spl image4 figure3

This is a table showing the primary endpoint events in demographic subgroups for a study that compared the effectiveness of Losartan Potassium and Atenolol in reducing stroke. The table includes different subgroups based on age, gender, race, pre-existing medical conditions, and other factors. The results indicate the rate of events in each subgroup, as well as the hazard ratio and confidence interval. The study adjusted for baseline Framingham risk score and level of electrocardiographic left ventricular hypertrophy.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.